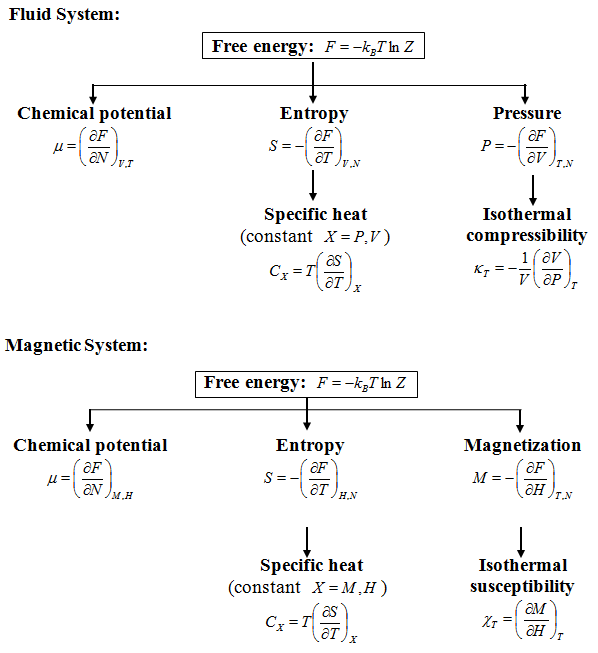
Thermal equilibrium corresponds to the minimum free energy or maximum entropy at finite temperature. All equilibrium thermodynamic properties can be calculated by taking appropriate derivatives of the free energy ![]() with respect to its parameters.
with respect to its parameters.
Note: For non-interacting system of N particles, the partition function Z can be written as ![]() where
where ![]() is the partition function for a single particle. Consequently one obtains ideal gas behaviour for
is the partition function for a single particle. Consequently one obtains ideal gas behaviour for ![]() .
.
Thermodynamic quantities can be calculated by taking different derivatives of the free energy F as given below: