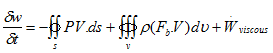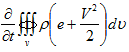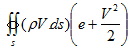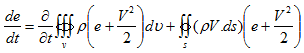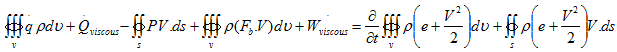4.1 Conservation of Energy (Integral Form)
The law of conservation of energy states that “energy can neither be created nor destroyed; it can only change its form”. Consider the CV shown in Fig. 2.1 as the thethermodynamic system. Let amount of heat δq be added to the system from the surrounding. Also let δw be the work done on the system by the surroundings. Both heat and work are the forms of energy. Addition of any form of the energy to the system, changes the amount of internal energy of the system. Lets denote this change of internal energy by de. From the principle of energy conservation.
δq + δw = de
Therefore in terms of rate of change the above equation changes to
or
|
(4.1) |
If the system to be considered as a open system then the change will take place for all the forms of energies owed by the system, like internal energy and kinetic energy. Hence right hand side of the above equation is just the representation of change in energy content of the system.
Lets first concentrate on first term on left hand side of above equation and evaluate the same. There are various sources of heat addition in the system some of which are external heat addition from the surrounding, heat addition by chemical energy release, heat addition by radiation etc. Lets consider some of those sources of heat addition and derive the energy equation. If ![]() is the amount of heat added per unit mass, then rate of heat addition for any elemental volume will be
is the amount of heat added per unit mass, then rate of heat addition for any elemental volume will be ![]() . Summing over the complete control volume gives us total external volumetric heat addition. Heat might get added by visous effects like conduction. Hence net rate of heat addition can be,
. Summing over the complete control volume gives us total external volumetric heat addition. Heat might get added by visous effects like conduction. Hence net rate of heat addition can be,
|
(4.2) |
Consider the second term on left hand side of equation (4.1) and evaluate the same. There are various ways by which work transfer can be achieved to or from the system. Main source is the surface forces like pressure, body force etc. Considering the elemental area ds of the control surface in Fig. 2.1. The pressure force on this elemental area is -Pds and the rate of work done on the fluid passing through ds with velocity V is (-Pds).V. Hence, summing over the complete control surface, rate of workdone due to pressure force is,
|
(4.3) |
In addition, consider an elemental volume dυ inside the control volume, as shown in Fig. 2.1. The rate of work done on the elemental volume due to body force is (ρFbdu).V. Here Fb is the body force per unit mass. Summing over the complete control volume, we obtain, rate of work done on fluid inside υ due to body forces is
|
(4.4) |
If the flow is viscous, the shear stress on the control surface will also do work on the fluid as it passes across the surface. Let ![]() denote the work done due to the shear stress. Therefore, the total work done on the fluid inside the control volume is the sum of terms given by (4.3) and (4.4) and
denote the work done due to the shear stress. Therefore, the total work done on the fluid inside the control volume is the sum of terms given by (4.3) and (4.4) and ![]() that is
that is
|
(4.5) |
Now consider the right hand side of equation (4.1) and eveluate the rate of internal energy change of the fluid. However since we are considering the open system we will have to consider the change in internal energy as well as the change in kinetic energy. Therefore right hand side of equation (4.1) should deal with total energy (sum of internal and kinetic energies) of the system. Let, e be the internal energy per unit mass of the system and kinetic energy per unit mass due to local velocity V be V2/2. Hence the rate of change of total energy is
|
(4.6) |
Total energy in the control volume might also change due to influx and outflux of the fluid. The elemental mass flow across ds is (ρV.ds). Therefore the elemental flow of total energy across the ds is (ρV.ds)(e+V2/2). Summing over the complete control surface, we obtain net rate of flow of total energy across control surface as,
|
(4.7) |
Hence the net energy change of the control volume is,
|
(4.8) |
Thus, substituting Equations (4.2), (4.5) and (4.8) into (4.1), we have
|
(4.9) |
This is the energy equation in the integral form. It is essentially the first law thermodynamics applied to fluid flow or open system.
4.2 One dimensional form of Conservation of Energy
Consider the control showin in Fig. (2.2) for steady inviscid flow without body force, Then the equation (4.9) reduces to,
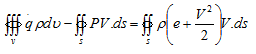
Let us denote the first term on left hand side of above equation by ![]() to represent the total external heat addition in the system. Thus, above equation becomes
to represent the total external heat addition in the system. Thus, above equation becomes
![]()
Evaluating the surface integrals over the control volume in Fig. 2.2, we obtain
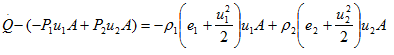
or
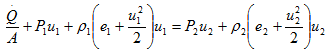
or
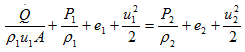
or
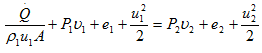
Here, ![]() /ρ1u1A is the external heat added per unit mass, q. Also, we know e + Pu = h. Hence, above equation can be re-written as,
/ρ1u1A is the external heat added per unit mass, q. Also, we know e + Pu = h. Hence, above equation can be re-written as,
|
(4.10) |
This is the energy equation for steady one-dimensional flow for inviscid flow.