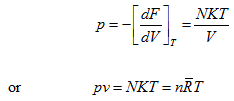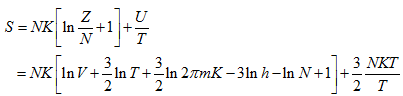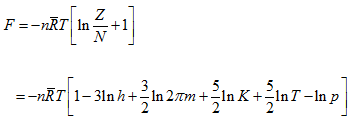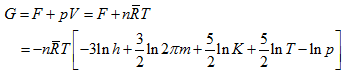Now,
|
(5.176)
(5.177) |
Which is the equation of state of an ideal gas.
Considering Entropy,
|
(5.178) |
Now,
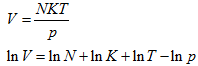
Substituting in Eq. 5.178,
|
(5.179) |
The molar entropy
|
(5.180) |
This is known as the Sackur-Tetrode equation for the absolute entropy of a monoatomic ideal gas. It is seen that
|
(5.181) |
where A stands for the sum of all terms that do not depend on T or V. This agrees with the equation from classical thermodynamics
|
(5.182) |
Also,
|
(5.183) |
and
|
(5.184) |
These are the absolute values of the properties of a monoatomic ideal gas.