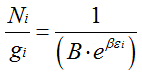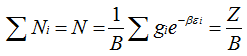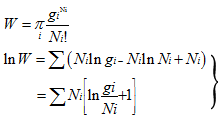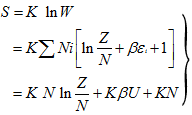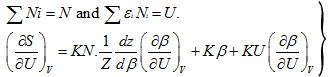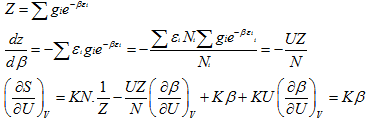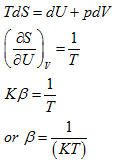Partition Function (z)
Both B-E and F-D statistics reduce to M-B statistics for Ni << gi. Gases follow M-B statistics except at cryogenic temperatures. The M-B distribution law for gases at equilibrium is
|
(5.143) |
For all the cells,
|
(5.144) |
The parameter, ![]() is called Partition Function or sum of state. This is an important parameter in statistical thermodynamics.
is called Partition Function or sum of state. This is an important parameter in statistical thermodynamics.
The partition function depends upon β and the way εi varies from cell to cell.
Substituting ![]() , the M-B distribution law becomes
, the M-B distribution law becomes
|
(5.145) |
It gives the most distribution of the molecules in a gas among the possible energy levels.
|
(5.146) |
Now,
|
(5.147)
(5.148) |
Thus
|
(5.149) |
Therefore,
|
(5.150) |
where
|
(5.151) |
Since
|
(5.152) (5.153)
(5.154) |
From
|
(5.155) |
The constant β is thus determined. Therefore, from Eq. (5.145)
|
(5.156) |
where
|
(5.157) |
The internal energy U of the system is
|
(5.158) |