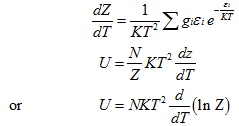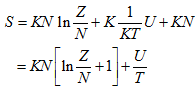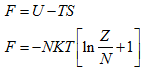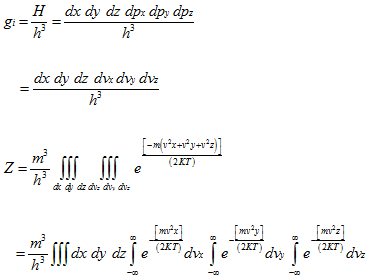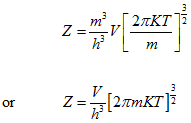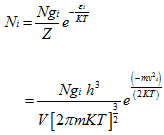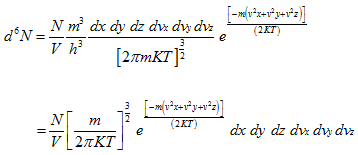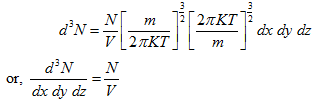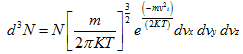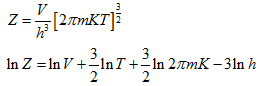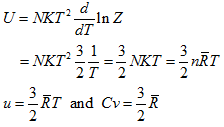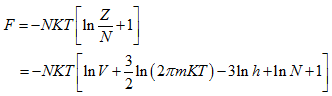Again,
|
(5.159) |
Entropy is given by
|
(5.160) |
The Helmholtz function is given by
|
(5.161) |
Once the partition function Z has been evaluated, all the thermodynamic properties can thus be estimated.
Monatomic Ideal Gas
The atoms of a monatomic ideal gas possess only translational kinetic energy. For an atom in cell i,
|
(5.162) |
For all the cells in the space occupied by the gas, the partition function is:
|
(5.163) |
where the degeneracy gi is given by
|
(5.164) |
Let I =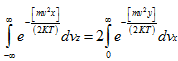
Assuming
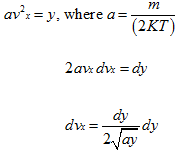
The integrand I is
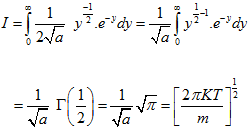
|
(5.166)
(5.167) |
Substituting in the equation,
|
(5.168) |
Substituting Ni by d6N,
|
(5.169) |
Integration of this equation over all values of vx, vy and vz gives
|
(5.170) |
The number of atoms per unit volume of ordinary space is thus a constant, confirming that the atoms are uniformly distributed in the gas volume.
The Eq. (5.169), when integrated over all the values of x, y and z, gives the distribution of atoms in velocity space, as given below:
|
(5.171) |
This equation is precisely the same as Maxwell-Boltzman velocity distribution as derived from the kinetic theory of gases and given by Eq. (21.49), provided K is recognized as the Boltzman constant.
Now,
|
(5.172)
(5.173) |
The internal energy of the gas from Eq. (5.159),
Thus, |
|
|
The results agree with the kinetic theory and the equipartition principle.
Considering the Helmholtz function
|
(5.175) |