Diffusion
Diffusion is a consequence of random molecular motion and occurs whenever
there is a concentration gradient of any molecular species in the system.
In other words, when the number of particles of one kind per unit volume on
one side of a surface differs from that on the other side there will be a
molecular diffusion. The phenomenon can be described as a transport matter,
(t molecules) across a surface. Diffusion can occur in gas, liquid as well
as solids. Present discussion is limited to diffusion of gas.
Some practical examples of diffusion are:
- Methods used to separate the isotopes U235 and U238. Naturally occurring uranium is converted to the hexafluoride UF6, a gas, and the mixture of isotopes flows by diffusion through a barrier.
- The operation of a nuclear reactor is also dependent on the phenomenon of diffusion. The neutrons in a reactor behave like a gas that is continuously being generated throughout the reactor by fission process and which diffuses through the reactor and eventually escapes from the surface. In order that the reactor may operate successfully, conditions must be such that the rate of generation of neutrons is at least as great as the loss by diffusion, plus the losses due to collisions in which the neutrons are absorbed.
Consider a vessel containing two different gases A and B separated by a partition
in the middle (Fig.nnn). Both the gases are maintained at the same pressure
and temperature. The number of molecules per unit volume is the same on both
sides. If the partition is removed, there is no large scale motion of the
gas in either direction, but after a sufficiently long time one can find that
both gases are uniformly distributed throughout the entire volume. The phenomenon,
as a result of which each gas gradually permeates the other is the diffusion.
The phenomenon of diffusion may be complicated by the fact that when more
than one type of molecule is present the rates of diffusion of one into the
other are not the same. We can simplify the problem and still bring out the
essential ideas by considering the diffusion of molecules of a single species
into others of the same species, known as self-diffusion.
If all of the molecules of a system were exactly alike, any calculation of
self-diffusion among them would be of academic interest only, since there
would be no experimental method by which the diffusing molecules could be
distinguished from others. However, molecules that are isotopes of the same
element, or molecules whose nuclei have been made radioactive differ only
in their nuclear (their mean kinetic energies will differ slightly because
of differences in Mass.) it is thus possible to “tag” certain
molecules so that they can be distinguished from others, and yet treat the
problem as if the molecules were all alike.
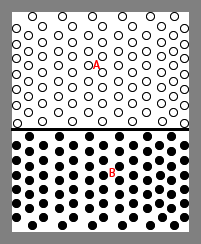
Fig. 4.21 A vessel containing two different gases separated by a partition
Consider an imagined horizontal surface S-S within the vessel of Fig. 4.21,
at some stage of the diffusion process. The vessel contains a mixture of tagged
and untagged molecules, the total number of molecules per unit volume being
the same at all points so that the pressure is uniform. We assume the temperature
to be uniform also. Let  represent the number
of tagged molecules per unit volume at any point. We shall assume that
represent the number
of tagged molecules per unit volume at any point. We shall assume that  is a function of y only, where the y-axis is normal to the surface S-S. If
is a function of y only, where the y-axis is normal to the surface S-S. If
 is positive, the downward flux of tagged molecules
across the surface is then greater than the upward flux. If
is positive, the downward flux of tagged molecules
across the surface is then greater than the upward flux. If ![]() represents the net flux of tagged molecules across the surface, per unit time
and per unit area, the coefficient of self –diffusion D is defined by
the equation
represents the net flux of tagged molecules across the surface, per unit time
and per unit area, the coefficient of self –diffusion D is defined by
the equation
|
(4.155) |
The negative sign is included since if  is
positive, the net flux
is
positive, the net flux ![]() is down-ward and negative.
is down-ward and negative.
We assume as before that each molecule makes its last collision before crossing
at a perpendicular distance 2l/3 away from the surface. If  is the number of tagged molecules per unit volume at the surface S-S, the
number per unit volume at a distance 2l/3 below the surface is
is the number of tagged molecules per unit volume at the surface S-S, the
number per unit volume at a distance 2l/3 below the surface is
|
(4.156) |
In the expression previously derived from the flux Φ, we must replace
n by  , and the upward flux
, and the upward flux  is then
is then
|
(4.157) |
In the same way, the downward flux is
|
(4.158) |
The net flux ![]() is the
difference between these, so
is the
difference between these, so
|
(4.159) |
Comparison with Eq. 4.151 shows
|
(4.160) |




