Let u0 represent the forward velocity of the gas at the plane G-G. At a distance 2l/3 above the surface, the forward velocity is
|
(4.139) |
Since the forward velocity gradient  can be considered
constant over a distance of the order of a free path. The forward momentum
of a molecule with this velocity is
can be considered
constant over a distance of the order of a free path. The forward momentum
of a molecule with this velocity is
|
(4.140) |
Hence the net momentum  in the direction of flow,
carried across the surface per unit time and per unit area by the molecules
crossing from above, is the product of momentum mu and the total flux Φ:
in the direction of flow,
carried across the surface per unit time and per unit area by the molecules
crossing from above, is the product of momentum mu and the total flux Φ:
|
(4.141) |
Similarly, the momentum carried across the surface by the molecules crossing from below is
|
(4.142) |
The net rate of transport of momentum per unit area is the difference between these quantities
|
(4.143) |
Hence, by comparison with the definition of the coefficient of viscosity in eq. (10-18) we have
|
(4.144) |
Interpretations:
- From the expression for viscosity derived (Eq. 4.144) it is found that viscosity of a gas is independent of pressure or density.
- Viscosity is a function of temperature as

- This theory does not hold good when the mean free path is in the dimension of the apparatus.
Table 4.3 values of the mean free path and molecular diameter of some gases determined from viscosity measurements. The values of l and d in this table were calculated using Eq.(10.13) for 1.

We can show that the mean speed  is given by
is given by
|
(4.145) |
So that
|
(4.146) |
Thus for molecules of a given species, the theory predicts that  is proportional to
is proportional to  , and that for different species
at a given temperature it is proportional to
, and that for different species
at a given temperature it is proportional to  .
.
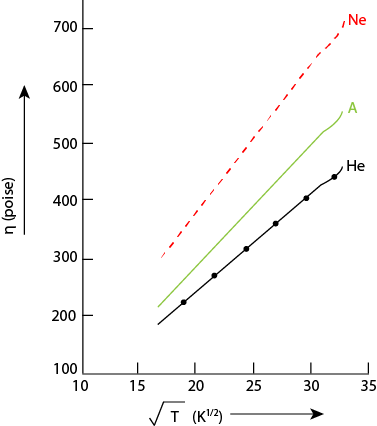
Fig. 4.20 Viscosity of Gases with Temperature
Figure 4.20 shows some experimental values of the viscosities of helium,
neon, and argon, plotted as a function of  . The
graphs are very nearly straight lines, but they curve upward slightly, indicating
that the viscosity increases with temperature at a somewhat greater rate than
predicted by the “hard-sphere” theory. This can be explained by
realizing that the molecules are not truly rigid spheres and that a “collision
“is more like that between two soft tennis balls than between two billiard
balls. The higher the temperature, the greater the average molecular kinetic
energy and the more the molecules become “squashed” in a collision.
Thus the centre to centre distance in a collision, and the corresponding collision
cross section , will be slightly smaller, the higher the temperature, with
a corresponding increase in η.
. The
graphs are very nearly straight lines, but they curve upward slightly, indicating
that the viscosity increases with temperature at a somewhat greater rate than
predicted by the “hard-sphere” theory. This can be explained by
realizing that the molecules are not truly rigid spheres and that a “collision
“is more like that between two soft tennis balls than between two billiard
balls. The higher the temperature, the greater the average molecular kinetic
energy and the more the molecules become “squashed” in a collision.
Thus the centre to centre distance in a collision, and the corresponding collision
cross section , will be slightly smaller, the higher the temperature, with
a corresponding increase in η.
As for the dependence of viscosity on the cross section σ, Eq.(4.146)
is as a matter of fact one of the relations used to “measure”
collision cross sections and the corresponding hard-sphere diameters d. Some
values of d computed from viscosity measurements, are given in Table 4.3