Coefficient of viscosity
Real gas is viscous in nature. This property can be illustrated with the kinetic
theory as discussed below:
Consider a real gas entrapped between two large plates separated by a distance
L as shown in Fig. 4.18

Fig. 4.18 Flow between two parallel plates
Because of the viscosity of the gas, a force F must be exerted on the upper plate to drag it to the right at constant velocity relative to the lower stationary plate. The molecules in the layer of gas have a forward velocity component which increases uniformly with the vertical distance y above the lower plate. The coefficient of viscosity of the gas, μ, is defined by the equation
|
(4.136) |
where A is the surface area of either plate and  is the velocity gradient at right angles to the plates.
is the velocity gradient at right angles to the plates.
The forward velocity of the molecules is superimposed on their large random
velocities rendering the gas to be under non-thermodynamic equilibrium condition.
However, in most practical problems the random velocities are so much larger
than any forward velocity that we can consider the system to be in equilibrium
condition.
The dotted line G-G in Fig. 4.18 represents an imagined surface within the
gas at an arbitrary height y above the lower plate. Because of their random
motions, there is a molecular flux Φ across the dotted surface, both from
above as well as from below. It can be assumed that at its last collision
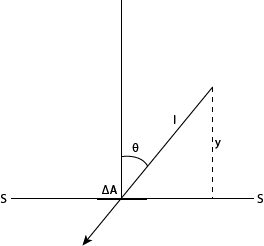
before crossing the surface (Fig. 4.15)
Fig. 4.19 Mean free path (y) before the molecule crosses the surface (S-S)
each molecule acquires a flow velocity towards the right, corresponding to
the particular height at which the collision was made. Since the flow velocity
above the dotted surface is greater than that below the surface, molecules
crossing from above transport a greater momentum towards the right across
the surface than do the molecules crossing from below. This results a net
rate of transport of momentum across the surface. From Newton’s second law
of motion we can equate the net rate of transport of momentum to the viscous
force (per unit area for both the quantities).Thus the viscosity of a gas
arises not from any “frictional” forces between its molecules, but from the
fact that they carry momentum across a surface as a result of their random
motion.
The molecules arriving at the surface G-G will on average have started their
last free paths before reaching the surface at a distance l away from the
surface. The perpendicular distance y from the surface for any θ - molecule
is y = l cosθ. The average value of y is found by multiplying l cosθ
by the flux ΔΦθ, summing over all values of θ
and dividing by total flux Φ. Thus
|
(4.137) |
Moreover, 
Therefore replacing Δθ with dθ and integrating over θ from zero to π/2
|
(4.138) |
Hence on the average, a molecule crossing the surface makes its last collision
before crossing at a distance equal to two-thirds of a mean free path above
(or below) the surface.