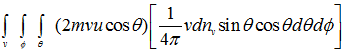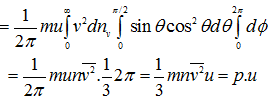Therefore, the loss of kinetic energy in all collisions per unit area and per unit time is:
|
(4.41) |
If the area of the moving piston is A, the loss of the molecular K.E. in all collisions on the piston per unit time is
|
(4.42) |
It is thus proved that when a gas expands adiabatically against a moving piston, work is done at the expense of the K.E. of the molecules, and hence the temperature of the gas decreases.