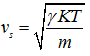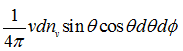where R = characteristic gas constant ![]()
Therefore, sonic velocity in an ideal gas
|
(4.36) |
which is somewhat smaller than the rms molecular speed.
Collision with a Moving Wall
Let us consider a gas expanding against a moving piston in a cylinder (Fig.4.5).
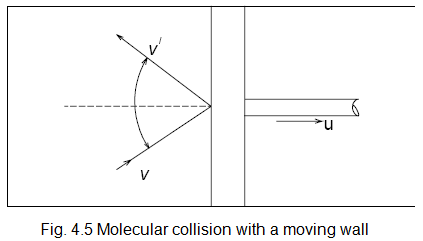
Let the piston move very slowly at a velocity u so that the gas remains in an equilibrium state at any instant, and the process becomes quasi-static and reversible. When a molecule collides elastically with a stationary wall, the magnitude of the normal component of velocity remains unchanged. If the wall is moving, the magnitude of relative velocity remains unchanged. If the normal component of the velocity before collision is vcosθ, the relative velocity is vcosθ - u. After collision the relative velocity is ![]() Therefore,
Therefore,
|
(4.37) |
or,
|
(4.38) |
The loss of Kinetic energy in the collision is
|
(4.39) |
Since u << v, the term 2mu2 is neglected. The number of collisions per unit area and per unit time is
|
(4.40) |