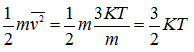From the expression obtain earlier for the pressure of an ideal gas (Eq 4.26), we can rewrite Eq 4.26 as
|
(4.28) |
where n = N / V. Again,
|
(4.29) |
where n = number of moles ,
![]() = universal
gas constant 8.3143 kJ(kgmol.K)
= universal
gas constant 8.3143 kJ(kgmol.K)
and T = absolute temperature.
Now,
n = N / N0,
where N0 is the Avogadro’s number ![]() molecules/kgmol. Therefore, the ideal gas equation of state is
molecules/kgmol. Therefore, the ideal gas equation of state is
|
(4.30) |
where K = Boltzmann constant = 1.38 X 10-23 J/(molecules.K). From
|
(4.31) |
|
(4.32) |
or,
|
(4.33a) |
where vrms is called the root-mean-square (rms) velocity of molecules, which is a function of temperature. Now,
|
(4.33b) |
The mean translational K.E. of a molecule is proportional to the absolute temperature, or conversely, the absolute temperature of gas is a measure of the K.E. of the molecules. The total translational K.E. of molecules, U, is
|
(4.34) |
The speed of a pressure pulse in an ideal gas is given by
|
(4.35) |