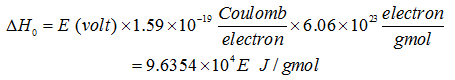where εε is the equilibrium value of the degree of ionization. This is known as the Saha’s equation. For a particular gas the degree of ionization increases with an increase in temperature and a decrease in pressure. It can be shown that ΔH0 is the amount of energy necessary to ionize 1 gmole of atoms. If we denote the ionization potential ΔH0 of the atom in volts by E, then
|
(3.71) |
Equation (3.68) becomes
|
(3.72) |
Expressing p in atmospheres, changing to common logarithms and introducing the values of B from statistical mechanics, Saha finally obtained the equation
|
(3.73) |
where ωi, ωe and ωa are constants that refer to the ion, electron and atom respectively. The value ωe for an electron is 2. The value of E, ωi, and ωa for a few elements are given below
Table 3.2 Values for E and ω
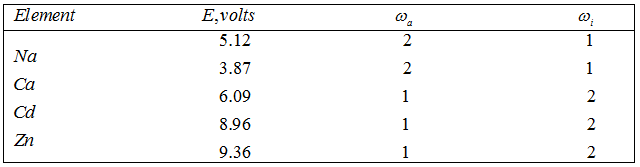
etc., the ionization potential is less. It means less energy is required to ionize one gmole of atoms. So these are used as seed for magnetohydrodynamic power generation. Saha applied Eq. (3.68) to the determination of temperature of stellar atmosphere. The spectrum of a star contains lines which originate from atoms (arc lines) and those which originate from ions (spark lines). A comparison of the intensities of an arc line and a spark line from the same element gives a measure of εε. Considering a star as a sphere of ideal gas, the pressure of a star can be estimated. Thus, knowing εε, p, E and the ω's, temperature of star can be calculated.