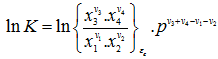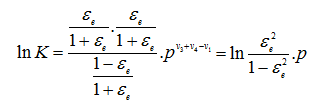Thermal Ionization of a Monatomic Gas
One interesting application of Nernst’s equation was made by Dr. M.N. Saha to the thermal ionization of a monatomic gas. If a monatomic gas is heated to a high enough temperature (5000 K and above), some ionization occurs, with the electrons in the outermost orbit being shed off, and the atoms, ions, and electrons may be regarded as a mixture of three ideal monatomic gases, undergoing the reaction
|
(3.65) |
Starting with n0 moles of atoms,
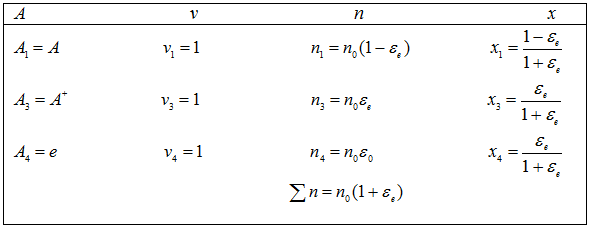
The equilibrium constant is given by
|
(3.66) |
or,
|
(3.67) |
Since the three gases are monatomic,![]() which, on being substituted in the Nernst’s equation, gives
which, on being substituted in the Nernst’s equation, gives
|
(3.68) |
where ![]()
Now,
|
(3.69) |
∴ |
(3.70) |