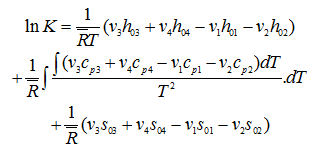Temperature Dependance of the Heat of Reaction
ΔH = v3h3 + v4h4 - v1h1 + v2h2 |
(3.56) |
h = h0 + ∫Cp dt |
(3.57) |
Therefore,
ΔH = v3h03 + v4h04 - v1h01 + v2h02 + ∫(v3Cp3 + v4Cp4 - v1Cp1 + v2Cp2)dT |
(3.58) |
Denoting ΔH = v3h03 + v4h04 - v1h01 + v2h02
ΔH = ΔH0 + ∫(v3C3 + v4C4 - v1C1 + v2C2)dT |
(3.59) |
If Cp is known as a function of temperature and if at any temperature ΔH is known, then at any other temperature, ΔH can be determined for a certain chemical reaction from the above relation.
Some chemical reactions may be expressed as the result of two or more reactions. If ΔH0 is known for each of the separate reactions, then ΔH0 of the resultant reaction may be calculated.
For example,

TEMPERATURE DEPENDENCE OF THE EQUILIBRIUM CONSTANT
ln K = -(v3Φ3 + v4Φ4 - v1Φ1 - v2Φ2) |
(3.60) |
where
|
(3.61) |
On substitution
|
(3.62) |
If
ΔH0 = v3h03 + v4h04 - v1h01 + v2h02 |
(3.63) |
Then,
|
(3.64) |
This equation is sometimes called as the Nernst’s equation.