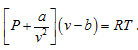Example 2:
Prove that heat at constant pressure is given by
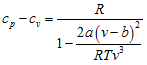
Solution:
the specific heat at constant pressure is given by

The specific heat at constant volume is given by
![]()
Therefore,
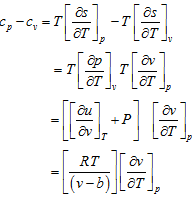
Differentiating van der Waals’ equation
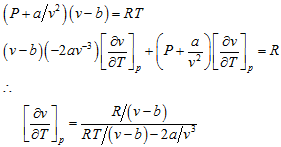
Substituting this value in the specific heat difference equation above,
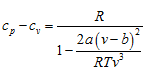
Example 3:
Joule-Thomson expansion apply to a van der Waals equation
For 1 mol of a real gas, the vander Waals equation takes the form
|
(C) |
Where a and b are constants. Transposing
|
(D) |
Or
|
(E) |
Now, a and b are very small. So their product ab is very small compared to v2 and as such ![]() term can be neglected, whereupon the foregoing equation simplifies to
term can be neglected, whereupon the foregoing equation simplifies to
|
(F) |