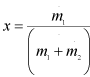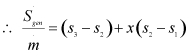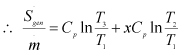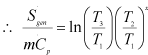Mass balance:
m1 + m2 = m3 = m(say) |
1.237 |
Let,
|
1.238 |
By first law of thermodynamics,
m1h1 + m2h2 = (m1 + m2)h3 |
1.239 |
or,
xh1 + (1 - x)h2 = h3 |
1.240 |
Since,
h = h(T) |
1.241 |
From Eq. (1.240)
xT1 + (1-x)T2 = T3 |
1.242 |
|
1.243 |
where
|
1.244 |
By second law of thermodynamics,
|
1.245 |
|
1.246 |
|
1.247 |
|
1.248 |
or,
|
1.249 |
Let, Ns Entropy generation number = ![]()
Now, substituting ![]() from Eq. (1.243) in Eq. (1.249), we get
from Eq. (1.243) in Eq. (1.249), we get
|
1.250 |
|
1.251 |
For, x = 1, Ns = 0 ⇒ m2 = 0, the system is with single stream.
For, ![]() , Ns = 0, temperature of both the streams are same.
, Ns = 0, temperature of both the streams are same.
Rate of exergy loss due to mixing is,
Wlost = I = T0Sgen |
1.252 |
|
1.253 |