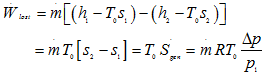(B) Quality degradation for a flow with friction
Consider a steady and adiabatic flow of an ideal gas through the segment of a pipe. Applying the first law of thermodynamics between sections (1)-(1) and (2)-(2) (Fig. 1.28)
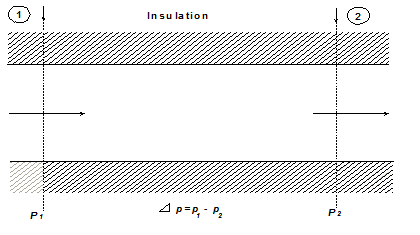
Fig. 1.28 Adiabatic steady flow of an ideal gas in a straight pipe
h1 = h2 |
1.231 |
Applying T-dS relationship
Tds = dh - vdp |
1.232 |
or,
|
1.233 |
or,
|
1.234 |
|
1.235 |
where ![]() since
since ![]() and higher order terms are neglected. Hence, lost
work or irreversibility is
and higher order terms are neglected. Hence, lost
work or irreversibility is
|
1.236 |
Hence decrease in exergy is proportional to the pressure drop as well as mass flow rate.
(c) Mixing of two fluid streams
Two fluid streams 1 and 2 of an incompressible fluid or ideal gas mixing adiabatically at constant pressure as shown in Fig. 1.29.
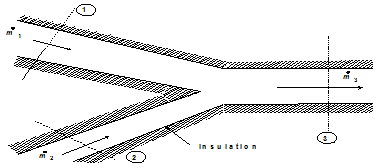
Fig. 1.29 Mixing of two fluid streams