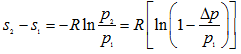Interpretations:
For the same heat loss at two different temperatures, exergy loss is more with higher temperature. For instance, exergy loss is more at 1000 K than that at 300 K for 1 kJ of heat loss.
The more the temperature, the more is the quality of energy. For example, quality of energy of a gas at 1000 K is superior to that at 400 K, since the gas at 1000 K has the capacity of doing more work than the gas at 400 K.
Some more examples of quality degradation
(a) Throttle Process
For frictionless, steady flow process of an ideal gas between sections (1)-(1) and (2)-(2) in a pipe (Fig. 1.27),
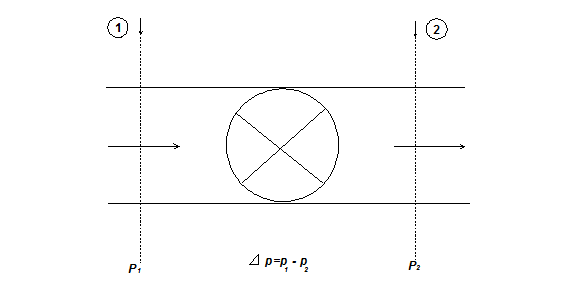
Fig. 1.27 Throttled flow through a pipe
h1 = h2 |
1.224 |
Again,
Tds = dh - vdp |
1.225 |
![]() and pv = RT
and pv = RT
|
1.226 |
or,
|
1.227 |
or,
|
1.228 |
|
1.229 |
|
1.230 |
Equation (1.230) is the expression for the irreversibility or lost work in the system comprising of the sections (1)-(1) and (2)-(2). It is obvious from the expression that quality degrades logarithmically with pressure drop between the sections as well as with the mass flow rate for an initial pressure p1 and surrounding temperature T0.