Demonstration of quality of energy based on exergy
Consider a hot gas of mass m flowing through a pipeline (Fig. 1.26). Due to the heat loss to the surroundings, the temperature of the gas decreases continuously from the inlet state 'a' to the exit state 'b'. Thus, the process is irreversible. However, for sake of simplicity, let us consider the process to be reversible and isobaric between the inlet and outlet. For an infinitesimal reversible process at constant pressure,
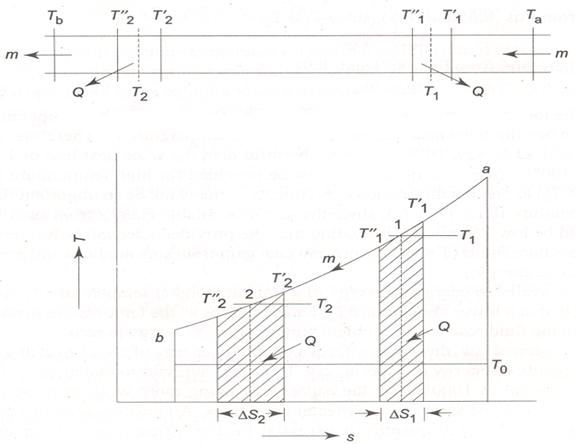
Fig. 1.26 Energy quality degrades along the flow direction
|
1.215 |
or,
|
1.216 |
Thus the slope ![]() depends on temperature T. With increase in T,
depends on temperature T. With increase in T, ![]() increases and vice versa.
increases and vice versa.
Let, Q unit of heat lost to the surroundings as temperature of gas decreases from ![]() to
to ![]() , T1 being average of the two temperatures.
, T1 being average of the two temperatures.
Heat loss at section 1-1
|
1.217 |
Exergy lost with this heat loss at temperature T1 is
W1 = Q - T0ΔS1 = T1ΔS1 - T0ΔS1 = (T1 - T0) ΔS1 |
1.218 |
At section 2-2, let heat loss be same as in section 1-1, Q(T2 < T1)
|
1.219 |
Exergy loss due to this heat loss at temperature T2 is
W2 = Q - T0ΔS2 = (T2 - T0) ΔS2 |
1.220 |
Considering the exergy loss at temperatures T1 and T2
W1 = Q - T0ΔS1 = T1ΔS1 - T0ΔS1 = (T1 - T0) ΔS1 |
1.221 |
W2 = Q - T0ΔS2 = (T2 - T0) ΔS2 |
1.222 |
As T1 > T2 , ΔS1 < ΔS2
∴ W1 > W2 |
1.223 |