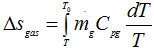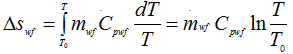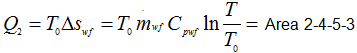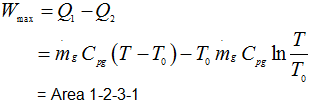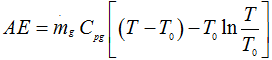Available energy from a finite energy source
Consider hot gas of mass mg at temperature T when the temperature of environment is T0 (Fig 1.25). Let the gas be cooled at constant pressure to T0 from state 1 and the heat given up by the gas Q1 is utilized in heating up reversibly a working fluid of mass mwf from state 3 to 1 along the same path so that the temperature difference between the gas and working fluid at any instant is zero and hence entropy increase of the universe is also zero.
The working fluid expands reversibly and adiabatically in an engine or turbine from state 1 to 2 doing work WE and then rejects heat Q2 reversibly and isothermally to return to the initial state 3 to complete the heat engine cycle.
Here,
Q1 = mgCpg(T - T0) = mwf Cpwf (T - T0) = Area 1-4-5-3-1 |
1.207 |
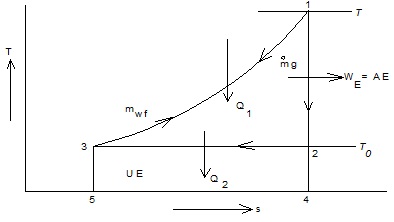
Fig. 1.25 Available and unavailable energy in finite processes
∴ mg Cpg = mwf Cpwf |
1.208 |
Now
( negative as T0 < T) |
1.209 |
|
1.210 |
∴ Δsuniv = Δsgas + Δswf = 0 |
1.211 |
|
1.212 |
∴ Available energy
|
1.213 |
Hence, exergy of a gas of mass mg at temperature T is
|
1.214 |