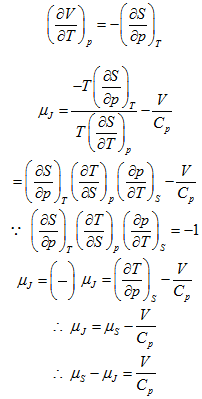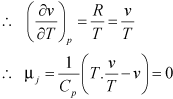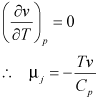For an ideal gas:
pv = RT |
1.195 |
|
1.196 |
There is no change in temperature when an ideal gas is made to undergo a Joule-Thompson expansion.
For an incompressible fluid:
|
1.197 |
Thus an incompressible fluid is heated while undergoing throttling process.
Application of Joule-Thompson effect:
- Vapor compression refrigeration system
- Production of dry ice or solid CO2 for low temperature refrigeration
Example:
Joule-Kelvin coefficient µj is a measure of the temperature change during a throttling process. A similar measure of temperature change produced by an isentropic change of pressure is provided by the coefficient µs where
![]()
Prove that: ![]()
Solution: The Joule –Kelvin coefficient µj is given by
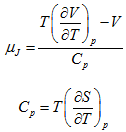
Maxwell's Relations