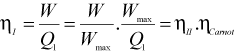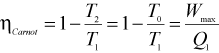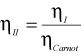Now,
|
1.27 |
where ηcarnot is the Carnot efficiency of an engine operating between two fixed temperatures. Efficiency of any thermodynamic cycle cannot be more than the Carnot cycle efficiency.
|
1.28 |
Hence,
|
1.29 |
Thermodynamics Processes
Second law of thermodynamics enables us to divide all the processes into two classes:
- Reversible or ideal process
- Irreversible or natural process
A process is reversible if, after the process has been completed, means can be found to restore the system and its elements of its and the surroundings to their respective initial states. Some of the reversible processes are:
|
Any process, which is not reversible, is irreversible. All natural (spontaneous) processes are irreversible. Some of the irreversible processes are:
|
Various processes can be expressed mathematically. The first as well as the second law can be combined together to analysis a thermodynamic system/process. Such mathematical relationships are elaborately discussed in Module 1 - Lecture 6.