Thermodynamic Properties
Both the first and second law of thermodynamics while applied to processes give rise to some thermodynamic properties. First law of thermodynamics while applied to a process for a closed system give rise to a property called internal energy U.
If Q is the amount of heat transferred to the system and W is the amount of work transferred from the system during the process (Fig. 1.9(a)-(b)), the net energy transfer (Q - W) will be stored in the system.
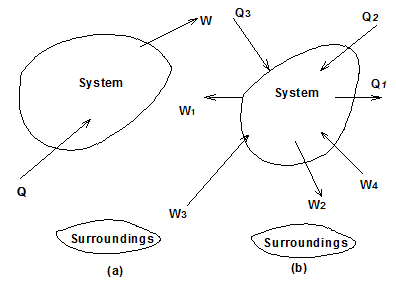
Fig. 1.9 (a)-(b) Thermodynamic system and surrounding
As both the heat and work are energies in transit, they can not be stored. Hence, the quantity (Q - W) is not either heat or work, but it is entirely a new form of energy, known as internal energy or simply the energy of the system.
∴ Q - W = ΔE |
1.3 |
where ΔE is the increase in energy of the system.
or,
Q = W + ΔE |
1.4 |
If there are more such energy transfer quantities involved in the process, as shown in Fig.1.9(b), the first law gives,
Q2 + Q3 - Q1 = ΔE + (W2 + W3 - W1 - W4) |
1.5 |