Tools for exergy analysis
The following are the tools for exergy analysis
- Value/Grassman diagram
- Pinch point technology
In general, conversion of energy or production of chemical substances in a plant requires determination of all the exergy flows that are transferred between distinguished apparatuses or unit operations of the plant. The resulting exergy losses can provide useful information with regard to the overall performance of the plant. However, it is generally difficult to judge the thermodynamic losses without any reference. Evaluation of plant performance will usually require a comparison of the thermodynamic performance of specific apparatuses or unit operations with available data from previously built plants. Thus, exergy efficiencies have appeared to be more useful, for large plants or integrated plants. It bears no significance to components. Exergy efficiencies combined with exergy flow diagrams can provide the thermodynamic performance of even for a complex system.
Q - T Diagram
Consider the heat transfer between two fluids in a recuperative type of heat exchanger. Temperature versus heat flow(T - Q) between the two fluids has been presented in Fig. 1.32.
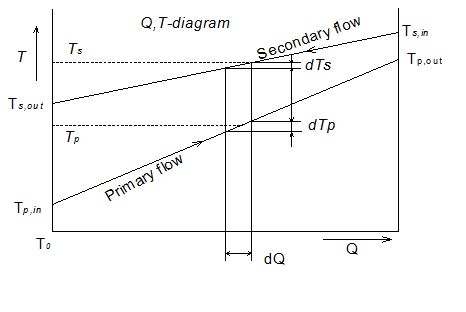
Fig. 1.32 Heat transfer process represented in a Q-T diagram
This diagram may be more informative if modified as follows:
Abscissa: indicates the heat transfer between the two streams
Ordinate: temperature T is replaced by the term ![]() . At , T = T0 the ordinate is zero and at T = α, ordinate value is unity (Fig. 1.33).
. At , T = T0 the ordinate is zero and at T = α, ordinate value is unity (Fig. 1.33).
exergy loss of heat transfer :
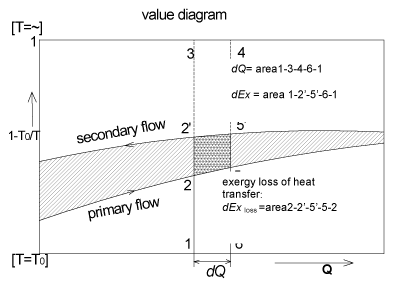
Fig. 1.33 Heat transfer process represented in a value diagram
If it is assumed that an infinitesimal small amount of heat dQ is transferred from the secondary flow, the flow that is cooled down in the heat exchanger, the resulting decrease in temperature dTs may be neglected. For the energy of this amount of heat can be written as
|
1.271 |