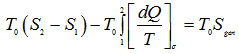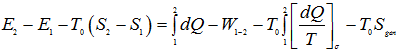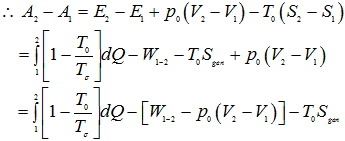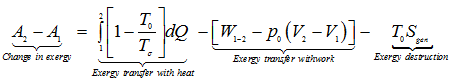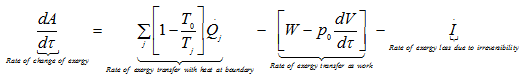Exergy balance for closed and open system
Exergy balance for the closed and open systems can be used to determine the locations, types and magnitude of losses of potential energy resources (fuels) and ways can be found to reduce such losses for making the energy system more efficient.
Closed system
For a closed system (Fig. 1.30), exergy or availability transfer occurs through heat and work interactions. No mass is transferred across the system boundary.
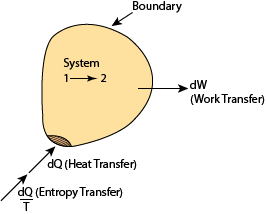
Fig. 1.30 A closed system
Ist Law of thermodynamics:
|
1.254 |
2nd Law of thermodynamics:
|
1.255 |
or,
|
1.256 |
Subtracting Eq. (1.256) from Eq. (1.254), we get,
|
1.257 |
Now, Defining availability function as
A = E + p0V- T0S |
1.258 |
|
1.259 |
|
1.260 |
In the form of rate equation,
|
1.261 |
where, Tj instantaneous temperature at the boundary
![]() rate of change of system volume.
rate of change of system volume.
For an isolated system,
A2 - A1 = ΔA = -I |
1.262 |
Since, I > 0, the only processes allowed by the second law of thermodynamics are those for which the exergy of the isolated system decreases. In other words,
The exergy of an isolated system can never increase.
(Counterpart of entropy principle, which states that entropy of an isolated system can never decrease.)