The reactions on each electrode of the battery are shown in Fig. 2. In the upper part of the diagram the battery is discharging. Both electrode reactions result in the formation of lead sulphate. The electrolyte gradually loses the sulphuric acid, and becomes more dilute.
When being charged, as in the lower half of Figure 2 , the electrodes revert to lead and lead dioxide. The electrolyte also recovers its sulphuric acid, and the concentration rises.
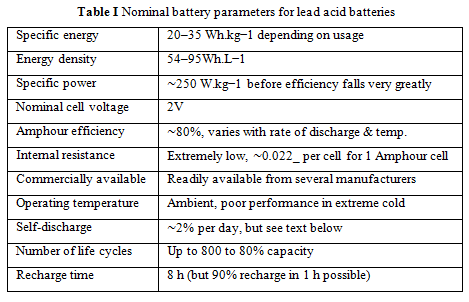
The lead acid battery is the most commonly used rechargeable battery in anything but the smallest of systems. The main reasons for this are that the main constituents (lead, sulphuric acid, a plastic container) are not expensive, that it performs reliably, and that it has a comparatively high voltage of about 2V per cell. The overall characteristics of the battery are given in Table I .
The figure given in Table I of 0.022 Ω per cell is a rule of thumb figure taken from a range of good quality traction batteries. A good estimate of the internal resistance of a lead acid battery is thus:
|
...........................................................(4) |
Table I Nominal battery parameters for lead acid batteries
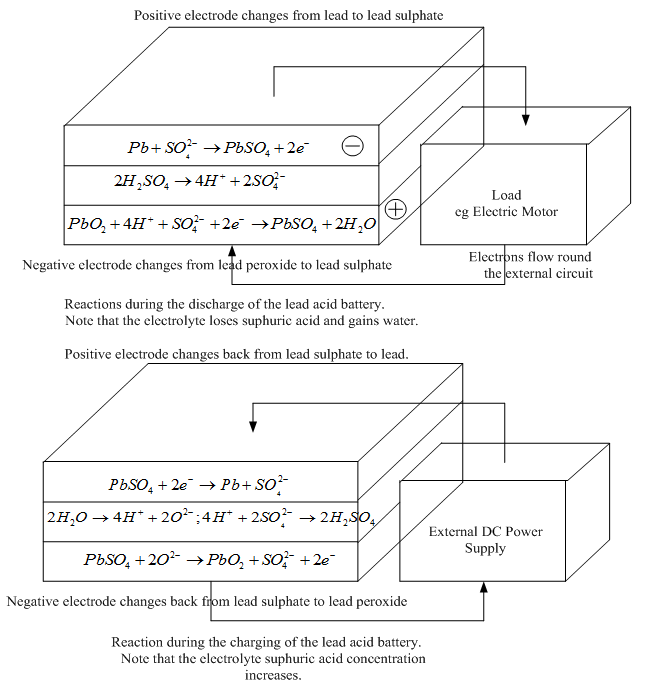
..................Fig. 2 The reactions during the charge and discharge of the lead acid battery
- Battery charging
Charging a lead acid battery is a complex procedure and, as with any battery, if carried out incorrectly it will quickly ruin the battery and decrease its life. As we have seen, the charging must not be carried out at too high a voltage, or water loss results.
There are differing views on the best way of charging lead acid batteries and it is essential that, once a battery is chosen, the manufacturer's advice is sought.
The most commonly used technique for lead acid batteries is called multiple steps charging. In this method the battery is charged until the cell voltage is raised to a predetermined level. The current is then switched off and the cell voltage is allowed to decay to another predetermined level and the current is then switched on again.