A battery consists of two or more electric cells joined together. The cells convert chemical energy to electrical energy. The cells consist of positive and negative electrodes joined by an electrolyte. It is the chemical reaction between the electrodes and the electrolyte which generates DC electricity. In the case of secondary or rechargeable batteries, the chemical reaction can be reversed by reversing the current and the battery returned to a charged state.
The ‘lead acid' battery is the most well known rechargeable type, but there are others. The first electric vehicle using rechargeable batteries preceded the invention of the rechargeable lead acid by quarter of a century, and there are a very large number of materials and electrolytes that can be combined to form a battery. However, only a relatively small number of combinations have been developed as commercial rechargeable electric batteries suitable for use in vehicles. At present these include lead acid, nickel iron, nickel cadmium, nickel metal hydride, lithium polymer and lithium iron, sodium sulphur and sodium metal chloride.
In this lecture the different types of the energy storage devices are presented. The following topics are covered in this lecture:
• Overview of Batteries
• Battery Parameters
• Lead acid batteries
• Lithium ion batteries
• Metal air batteries
• Battery Charging
Overview of Batteries
From the electric vehicle designer's point of view the battery can be treated as a ‘black box' which has a range of performance criteria. These criteria will include:
• specific energy
• energy density
• specific power
• typical voltages
• amp hour efficiency
• energy efficiency
• commercial availability
• cost, operating temperatures
• self-discharge rates
• number of life cycles
• recharge rates
The designer also needs to understand how energy availability varies with regard to:
• ambient temperature
• charge and discharge rates
• battery geometry
• optimum temperature
• charging methods
• cooling needs.
However, at least a basic understanding of the battery chemistry is very important, otherwise the performance and maintenance requirements of the different types, and most of the disappointments connected with battery use, such as their limited life, self-discharge, reduced efficiency at higher currents.
Battery Parameters
- Cell and battery voltages
All electric cells have nominal voltages which gives the approximate voltage when the cell is delivering electrical power. The cells can be connected in series to give the overall voltage required. The ‘internal resistance' and the equivalent circuit of a battery is shown in Figure 1 . The battery is represented as having a fixed voltage E , but the voltage at the terminals is a different voltage V , because of the voltage across the internal resistance R . Assuming that a current I is flowing out of the battery, as in Fig. 1, then by basic circuit theory we can say that:
V = E - IR |
.......................(1) |
..................................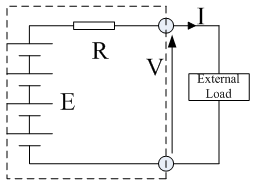
Fig. 1 Simple equivalent circuit model of a battery. This battery is composed of six cells