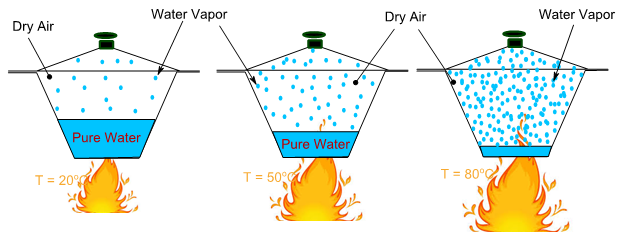
Fig. 2.13. Thermodynamic equilibrium between water and air at different temperatures
As the temperature increases, the presence of vapor in the air increases. Therefore, the air now contains water vapor as one of the components apart from other components of dry air discussed before. The water vapor and its exerted pressure increases exponentially with temperature. The vapor pressure reaches the atmospheric pressure at the boiling point temperature, 100°C. The pressure exerted by the water vapor in a unit volume of closed system at a given temperature can be calculated using the following Tetens' equation: (1930 : Uber einige meteorologische Begriffe. Zeitschrift fur Geophysik, Vol. 6:297 ) :
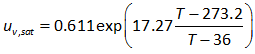 ........................................................(2.5)
........................................................(2.5)
where t is expressed in
degree Celsius. The following equation has also been used commonly to understand the dependency of saturated vapor pressure on temperature:
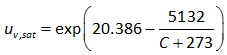 ..............................................................(2.6)
..............................................................(2.6)
Another noted equation for calculating uv,sat is based on the Antoine's equation(1888 : Tensions des vapeurs; nouvelle relation entre les tensions et les températures" [Vapor Pressure: a new relationship between pressure and temperature], Comptes Rendus des Séances de l'Académie des Sciences (in French) 107 : 681–684, 778–780, 836–837 ) :
![]() .............................................................................(2.7)
.............................................................................(2.7)