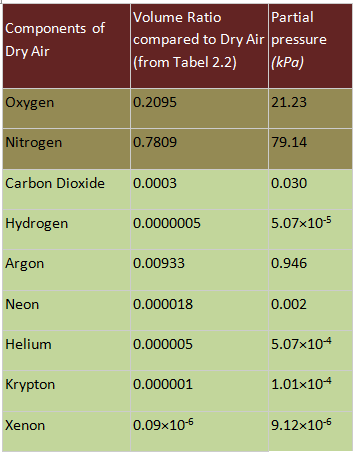
The partial pressures of different components of air are computed and presented in Table 2.3 considering the atmospheric pressure to be 101.345 kPa.
Table 2.3. Partial pressures of the components of dry air
(iii) all the components of air, including the water vapor, reach thermodynamic equilibrium. It enforces that the chemical potentials among all components of all phases in a given system is same.
Let us assume equilibrium between the free water and air in a closed container at a standard temperature 20°C as shown in the first picture in Fig. 2.13.