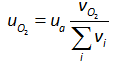A qualitative understanding of an important state variable, i.e. the relative humidity, which defines equilibrium between water and air phases requires the following assumptions to be valid (Lu and Likos, 2004).
(i) the composition of air, excluding the water vapor component, at a given spatial location remains essentially same over time as presented in Fig. 2.5 for the dry air . However, in humid conditions, the atmospheric air contains a water vapor component which may significantly vary from space and time under the dynamic variables of the atmosphere. In unsaturated soils, the water vapor component is understood to be the most important component of the air which changes the overall “chemical” composition of the soil pore air. The prevailing amount of water vapor present in equilibrium with the soil is often used in the measurement of important state variable.
(ii) all the gas components follow the ideal gas behavior . Therefore, the partial pressure of each air component can be obtained from the molar fraction of each component as shown below
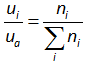 .......................................................................................................(2.3)
.......................................................................................................(2.3)
where the subscripts “ i ” denotes the component of air, n denotes the molar fraction, and the ua is the atmospheric air pressure. The Eq. (2.3) can be written in terms of volume fraction as ideal gas has the constant volume, which is equal to 22.4 L/mol. Therefore, the Eq. (2.3) can written as
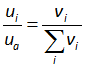 .....................................................................................................(2.4)
.....................................................................................................(2.4)
where v denotes the volume fraction of the air. The partial pressure of oxygen can, then, be calculated using the following equation if the volume fraction of oxygen in the atmospheric air and the atmospheric pressure of air are known: