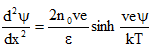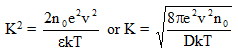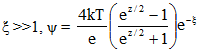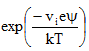In this model, double layer is represented by negatively and positively charged sheets of equal magnitude (Yong 2001). In this model, positive charges are considered to be stationery, which is against the reality that cations are mobile. It is opined that this model is too simple to address the real complexities of double layer.
B) Gouy Chapman model: Gouy suggested that interfacial potential at the charged surface can be attributed to the presence of a number of ions of given sign attached to the surface and to an equal number of opposite charge in the solution. The counter ions tend to diffuse into the liquid phase, until the counter potential set up by their departure restricts its diffusion. The kinetic energy of counter ions affects the thickness of resulting double layer. Gouy and Chapman proposed theoretical expression for electric potential in double layer by combining Boltzman equation (2.1) and Poisson equation (2.4), where in Eq. 2.1 relates ion distribution to electric potential and Eq. 2.4 relates electric potential and distance (Reddi and Inyang, 2000). This combination is given by Eq. 2.7. For the case of a single cation and anion species of equal valency (i=2) and n0 = n0+ = n0- and v+ = v- = v, then Eq. 2.7 simplifies to Poisson-Boltzmann equation (Eq. 2.8).
|
2.8 |
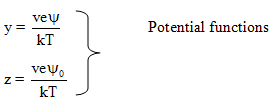
Solutions of the above are usually given in terms of the dimensionless quantities as stated below.
where |
|
D is the dielectric constant of the medium. According to Eq. 2.11, K depends on the characteristics of dissolved salt and fluid phase. However, actual values of concentration and potential at any distance from the surface would also depend on surface charge, surface potential, specific surface area and dissolved ion interaction. This means that the type of clays and pore solution are very important.
Solution can be obtained for a set of boundary conditions, one at the surface and other at infinite distance:
![]()
ψ0 is the potential at the clay surface.
For z<<1, ψ = ψ0e-Kx |
2.12 |
|
2.13 |
For some arbitrary z and |
2.14 |
Eq. 2.12 is commonly referred to as Debye-Huckel equation and 1/K represents characteristic length or thickness of double layer (Mitchell and Soga 2005). Knowing electric potential from above equations, it is possible to determine ion distribution from Eq. 2.3.
For cations: n+ = ni0 |
2.15 |
For anions: n- = ni0 |
2.16 |
This model is accurate only if the soil behaves like a true parallel particle system. It does not satisfactorily provide description of ψ immediately adjacent to the charged particle. This is mainly due to the mechanisms associated with chemical bonding and complexation. Gouy-Chapman model is ideally suited for qualitative comparisons. The basic assumption in Botlzmann equation where in the potential energy is equated to the work done in bringing the ion from bulk solution to some point, does not consider other interaction energy components.
C) Stern model
According to Stern model total cations required to balance the net negative charge on clay surface consists of two layers. The first layer is of cations are adsorbed on to the clay surface and are located within a distance of δ. The clay surface charges and the adsorbed group of cations are termed as electric double layer (EDL) or Stern layer. The other group of cations are diffused in a cloud surrounding the particle and can be described by Boltzmann distribution as discussed in the previous section. The total surface charge (σs) is counter balanced by Stern layer charge σδ; and diffuse layer charge σdl. The surface potential (ψs) depends on electrolyte concentration and surface charge (whether it is constant or pH dependent). It decreases from ψs to ψδ; when the distance increases from surface to the outer boundary of Stern layer. Beyond this distance, ψ is quantified by using Eq. 2.13.
There are other DDL models like DLVO which deals with complex interactions. However, these are not discussed in this course. The interested readers can refer to Yong (2001) for further reading.