Diffused double layer (DDL)
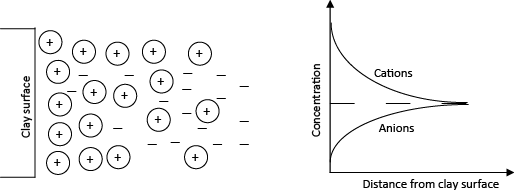
Diffused double layer (DDL) is the result of clay-water-electrolyte interaction. Cations are held strongly on the negatively charged surface of dry fine-grained soil or clays. These cations are termed as adsorbed cations. Those cations in excess of those needed to neutralize electronegativity of clay particles and associated anions are present as salt precipitates. When dry clays come in contact with water, the precipitates can go into solution. The adsorbed cations would try to diffuse away from the clay surface and tries to equalize the concentration throughout pore water. However, this movement of adsorbed cations are restricted or rather minimized by the negative surface charge of clays. The diffusion tendency of adsorbed cations and electrostatic attraction together would result in cation distribution adjacent to each clay particle in suspension. Fig. 2.8 presents such a distribution of ions adjacent to a single clay particle. The charged clay surface and the distributed ions adjacent to it are together termed as diffuse double layer (DDL). Close to the surface there is high concentration of ions which decreases outwards. Thus there are double layers of ions (a) compressed layer and (b) diffused layer and hence the name double layer. The variation in concentration of cations and anions in pore water with distance from clay surface is also presented in Fig. 2.8. A high concentration of cations close to clay surface gradually reduces, and reaches equilibrium concentration at a distance away from clay surface. For anions, concentration increases with distance from clay surface.
|
|
|
Several theories have been proposed for defining ion distribution in DDL. Gouy and Chapman is one of the initial explanations on DDL ion distribution (Mitchell and Soga 2005). The theory has been further modified by Derjaguin and Landau; Verwey and Overbeek which is known by the name DLVO theory (Mitchell and Soga 2005). In addition to ion quantification, DLVO describe the repulsive energies and forces of interaction between clay particles and prediction of clay suspension stability. Sposito (1989) observed that the theory predicts ion distribution reasonably for only smectite particles suspended in monovalent ion solution at low concentration. However, the theory can still be used for defining forces of interaction, flocculation, dispersion, clay swelling etc. A much more refined description of interparticle forces has been proposed by Langmuir (1938) and extended by Sogami and Ise (1984).
Following are the assumptions which pertain to the formulation of DDL theory:
- Ions in the double layer are point charges and there are no interactions among them.
- Charge on particle surface is uniformly distributed.
- Platy particle surface is large relative to the thickness of double layer (to maintain one dimensional condition).
- Permittivity of medium adjacent to particle surface is independent of position. Permittivity is the measure of the ease with which a molecule can be polarized and oriented in an electric field.
Concentration of ions (no of ions/m3) of type i, ni, in force field at equilibrium is given by Boltzmann equation as follows:
ni = |
2.1 |
E is the potential energy, T is the temperature in Kelvin, k is the Boltzmann constant (1.38 x 10M-23 J/K), subscript 0 represents reference state which is at a large distance from the surface.
Potential energy of an ion “i” in electric field is given by Eq. 2.2.