11.13.4 Oxidation
- Bromine water oxidizes aldehyde functional group, but it cannot oxidize ketones or alcohols. Therefore, aldose can be distinguished from ketose by observing reddish-brown colour of bromine. The oxidized product is an aldonic acid (Scheme 4).
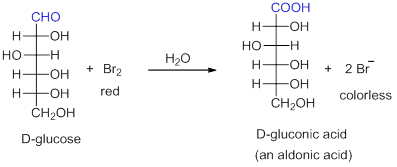
Scheme 4
- Tollen's reagent can oxidize both aldose and ketose to aldonic acids. For example, the enol of both D-fructose and D-glucose, as well as the enol of D-mannose are same (Scheme 5).
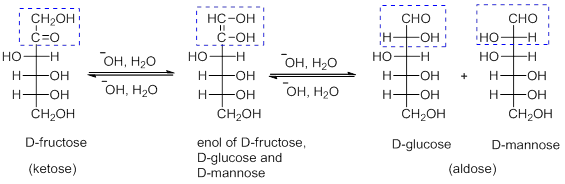
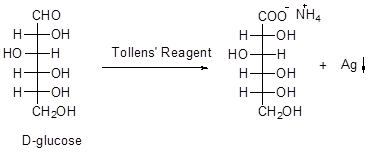
Scheme 5
Both aldehyde and primary alcohol groups of an aldose are oxidized by strong oxidizing agent such as HNO3. The oxidized product called an aldaric acid. Ketose also reacts with HNO3 to give more complex product mixtures (Scheme 6).
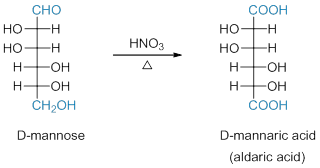
Scheme 6