Fajan´s Rule:
An ion will be formed most easily when ;
(i) the electronic structure of the ion is stable,
(ii) the charge on the ion is small, and
(iii) a small atom forms the anion and a large atom forms the cation.
Increased covalence is favored by ;
(i) high charge on the ions,
(ii) small size of the cation and large size of the anion,
(iii) cations with 18 electron structures, i.e. novel-gas configuration. Noble gas configuration of the cation produces better shielding and less polarizing power.
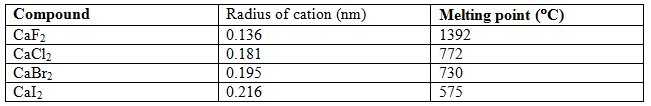
Examples: Development of covalent character with increase of cation charge (high charge on ions)
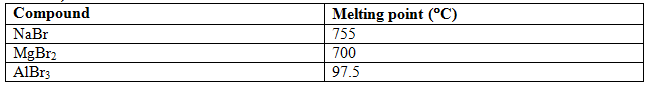
Examples: Decrease of covalent character with increase of cation radius
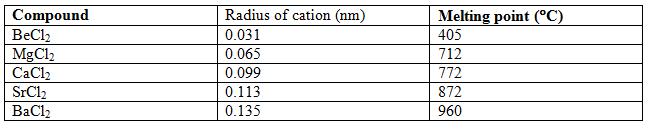
Examples: Development of covalent character with increase of anionic radius