n -Type semiconductor:
Both Ge and Si contain four electrons in the outer most shell, i . e . four valence electrons. Some atoms with five outer electrons, like arsenic (As), is added to Ge or Si crystals. This process is known as doping. When one Si or Ge atom will be replaced by a As atom, four out of five valence electros of As will be used for covalent bonding with other four Ge or Si atoms and the fifth one will remained unused. At room temperature some of the fifth electrons on As are excited in to the conducting band. Therefore, current can be carried out easily even at room temperature.This is called extrinsic conduction. As current is carried out by excess electron, the semiconductor in the presence of As is known as n -type semiconductor.
p -Type semiconductor:
When a material having outer shell electron one less than four [ e . g . Indium (In)] is doped into Si or Ge a p -type semiconductor is generated.
Each In uses three of its outer electrons for covalent bonding with Si or Ge to form a three bond lattice. As Si or Ge has four outer electrons, hence, one site remains vacant and a positive hole is created. With increase of temperature electron from Si or Ge may occupy the hole position. Consequently another hole is created. Such a way a movement of positive hole occurs and electrons movement, i . e . current flow, is opposite to the hole movement. Since current is carried by migration of positive hole, this type of semiconductor is called p -type semiconductor.
Solvation energy:
In a polar solvent an inherent dipole moment exist in the solvent molecules. Therefore, when an ionic solid is placed into a polar solvent each ion get surroundings by the opposite pole of the solvent molecule. This phenomenon is called salvation and the enthalpy change takes place by this salvation is known as salvation energy. Let us consider the solvent as water (H2O).
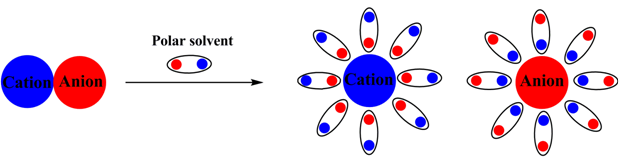
Figure 2.17. Schematic representation of salvation of an ionic solid.
When the salvation energy is more compared to the lattice energy of an ionic solid, the solid dissolves into the solvent. This indicates that the lattice will less lattice energy will be more and easily soluble in an ionic solid compared to the other which has more lattice energy.
Some Examples:
(1) Cesium iodide (CsI) has less lattice energy compared to sodium fluoride (NaF). Therefore, CsI is more soluble in water that that of NaF.
(2) CaF2 is not soluble in water due to extremely high lattice energy, while, CaCl2 is fairly soluble in water as its lattice energy is lower compared to CaF2 .
(3) BaO having less lattice energy is about 2000 times more soluble in water than that of MgO.
(4) The solubility in water decreases in the following order for AgF, AgCl, AgBr, AgI ;
AgF > AgCl > AgBr > AgI
Here, AgI has lower lattice energy that AgF. Still, it is less soluble in water. This is because of the increasing of covalent character in the compound on moving from AgF to AgI.
(5) The radious of barium ion (Ba2+) is almost double to magnesium ion (Mg2+). Hence, the lattice energy of BaSO4 should be lower than that of MgSO4 . Therefore, BaSO4 should be more soluble in water than MgSO4 . In fact, it is reverse. The reason is the large sulphate anion (SO42−). Small cation is less in contact with large anion. Therefore MgSO4 forms less compact lattice that BaSO 4 and consequently, less lattice energy and more solubility (easily soluble).
Polarization:
In a lattice, cation(s) is(are) surrounded by anion(s) and vice versa . The attraction force between them is coulomb attraction. When cations and anions in a lattice come to their equilibrium distance, the relatively loose electron cloud in the anion is attracted more by the positively charged cation. Therefore, the electron cloud around the anion undergoes deformation. This deformation is said to be polarized. The reverse polarization is extremely less as positively charged cations hold their electron cloud very tightly.
With increasing of polarization the charge cloud between cation and anion tends to mix or merge with each other. This extent of mixing determines the ionic and covalent character of a compound. Higher the mixing, higher the covalent character is.