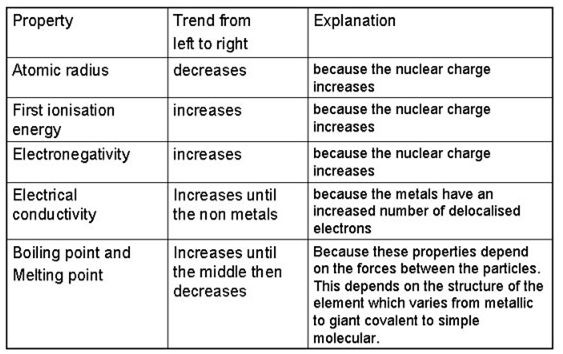
Atoms with stronger effective nuclear charge have greater electron affinity. Therefore, electron affinity from left to right in a period increases, while, in a group on moving from top to bottom, electron affinity decreases.
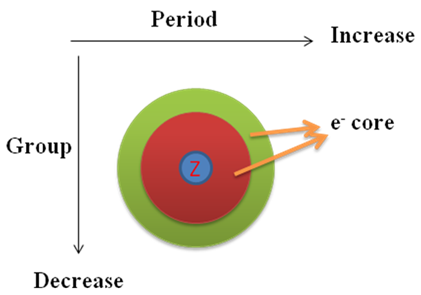
The 2 nd , 3 rd , and so on electron affinity is higher compared to 1 st as the extra electron causes extra repulsion between the incoming electron and the existing electron core in a atom.
The Electron affinity of the noble gasses are negative, indication their reluctance to accept an extra electron in their close, stable shell. On the other hand, the halogens posses highly positive electron affinity, which showing their high tendency to accept an electron to have a stable noble gas electronic configuration.
Electron affinity of Cl is greater than F. F is smaller in size and has high electron density compared to Cl. Due to these factors electron are repelled strongly in case of F hence the electron affinity of F is lower compared to Cl.
(F) Electronegativity:
Electronegativity is a measure of the attraction of an atom for the electrons in a chemical bond. This is unit less parameter.
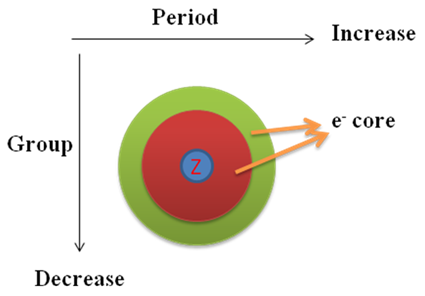
With increase of effective nuclear charge in an atom the power to draw electron pair of a chemical bond towards its own nucleus increases and hence, electro negativity increases. As the effective nuclear charge increases along left to right in a row, electronegativity increases in the same direction. In a group effective nuclear charge decreases from top to bottom so, electronegativity decreases from top to bottom.
Fluorine is the most electronegative element in the periodic table.
(G) Metallic character:
The metallic character of atoms can be related to the desire to lose electrons.
On the periodic table, the metallic character of the atoms increase down a family and decreases from left to right across a period.