(D) Ionization energy:
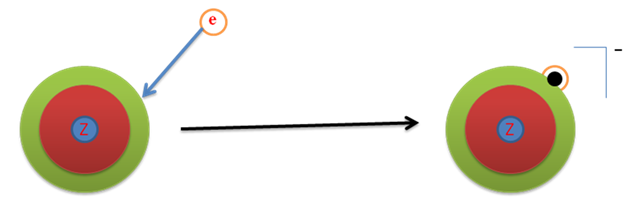
Defined as the amount of energy necessary to remove the outer most electron from an isolated neutral atom in the gaseous stat to infinity. This is also known as 1st ionization energy. Abbreviation is E i , it has units of kJ/mol.
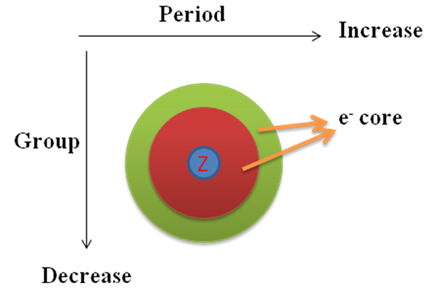
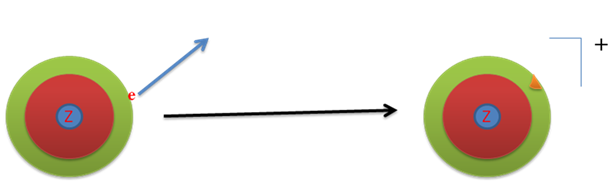
As the effective nuclear charge increases along a row from left to right, electrons are held more tightly from left to right. Therefore, the energy required to remove an electron, i . e . ionization energy will increase from left to right in a row.
Similarly, in a group on moving from top to bottom effective nuclear charge decreases, hence, electron are held more loosely on moving from top to bottom, consequently, ionization energy decreases.
The 1st ionization energy is always lower compared to 2 nd , 3 rd , so on, ionization energy. This is because after the removal of 1 st electron, the system becomes mono positive. Due to this reason electrons are more tightly hold by the nucleus. Therefore, more energy is required to remove electron further.
The 1st ionization potential of N or P is more compared to O or S.
In N the outer electronic configuration is 2 s 2 2 p 3 . The three electrons in 2p orbital are all in same spin and provide a stable configuration. In O the outer electronic configuration is 2 s 2 2 s 4 . In this case one of the three p orbitals occupies two electrons that are anti parallel to each other. Because of having two electrons in a same sub shell a repulsion between electron occurs which lower the electron releasing or ionization energy.
(E) Electron affinity:
Electron affinity reflects the ability of an atom to accept an electron. It is the energy change that occurs when an electron is added to a gaseous atom. This is also known as 1st electron affinity.
Abbreviation is E a , it has units of kJ/mol.