Effective nuclear charge (Z eff):
The s , p , d , and f orbitals within a given shell have slightly different energies.
The difference in energies between subshells results in electron–electron repulsion which shields outer electrons from the nucleus.
The net nuclear charge felt by an electron is called the effective nuclear charge (Z eff).
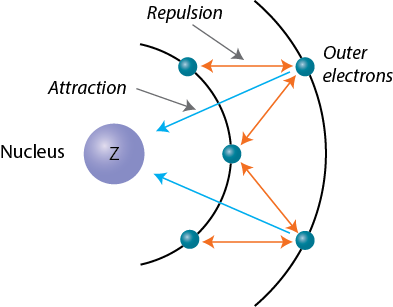
Figure1.17. Shielding mechanism.
Z eff is lower than actual nuclear charge.
Z eff = Z - σ (Shielding constant)
σ = 0.35 (for n )
= 0.85 (for n -1)
= 1 (for ≥n -2)
We know that Z eff increases toward nucleus
ns > np > nd > nf
(C) Atomic radii:
The atomic radius of an element is half of the distance between the centers of two atoms of that element that are just touching each other. Generally, the atomic radius decreases across a period from left to right and increases down a given group.
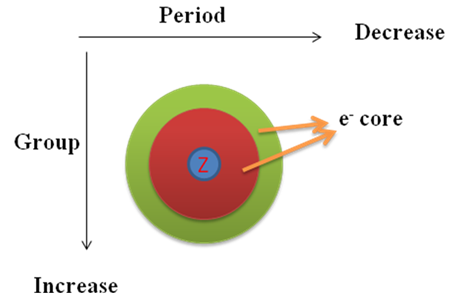
As we go along a period from left to right nuclear charge as well as number of electron increases successively. The additional electron occupies same orbital (let say red core). Therefore, the effective nuclear charge increases and electron are held more tightly and closely to the nucleus. This is why in a row on going from left to right size of atom decreases.
If we go down a group, additional electron occupies new orbital (let say green). Consequently, electrons are more screened by the inner core electron cloud. Hence, the effective nuclear charge on the electron cloud decreases on moving from top to bottom in a group, which causes an increase in atomic size.