Trivial names of group:
- Group 1: Alkali metals
- Group 2: Alkaline earth metals
- Group 11: Coinage metals
- Group 15: Pnictogens
- Group 16: Chalcogens
- Group 17: Halogens
- Group 18: Noble gases
IUPAC name of the elements:

Example (naming):
125 = umbipentium; ium comes always at the end
Trends in physical properties changes:
(A) Melting and boiling points:
A starts melting when then the thermal energy is more compared to the force holding their constituent unit in position is overcome. Boiling involves complete detachment of the closely packed constituent liquid elements. Therefore, both the properties correspond to the extent of inter - particle or inter - atomic bonding. There are several factors, like lattice energy, hydrogen bonding, packing, etc are related to the melting and boiling temperature. Though melting and boiling properties are controlled by several factors, periodical trends have been observed.
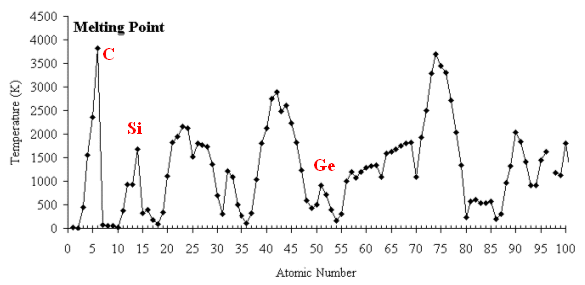
Figure1.14. Periodic variation of melting point of elements.
On going from left to right in a period, the melting temperature increases and it reaches at the highest point for the alkaline earth metals and then further decreases.
- Nobel gases are very low melting. The novel gas atoms contained filled electron shell. Therefore, inter - atomic binding is very low and they melt easily.
- The alkali metals have on one electron in their outer shell. Hence, inter - atomic binding is not so strong and that is way they do not have high melting temperature.
- The alkaline earth metals have two unpaired electrons and therefore, inter - atomic interaction in higher compared to the alkaline metals, and the alkaline earth metals are very gigh melting.
- Nitrogen, oxygen, and fluorine form inter - atomic triple, double, and single bond respectively. Because of high repulsion between non - bonding electrons or higher electron density around the atoms they acquire low melting point.
As we go down a group, the size of atom increases and hence, inter - atomic interaction also decreases. Therefore, melting temperature decreases.
Interestingly, the variation of melting point along a transition metal series is irregular. There are high extents of d orbital contribution to the melting point determining factors.