Modern periodic table:
In the modern period table elements are placed progressively according to their atomic number. The table is consists of 18 groups and 7 periods. The arrangement is presented by Bury (1921) based on electron distribution in a Bohr-Sommerfeld atom model and also known as the Bohr table.
Table. Modern (Bohr) periodic table*:
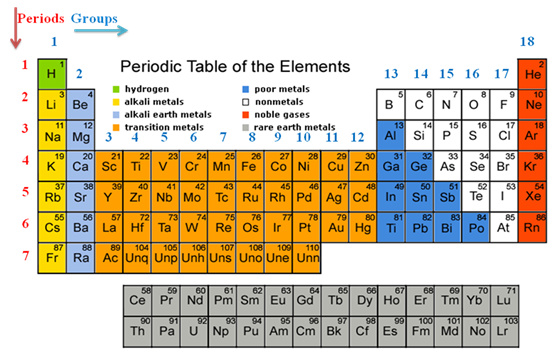
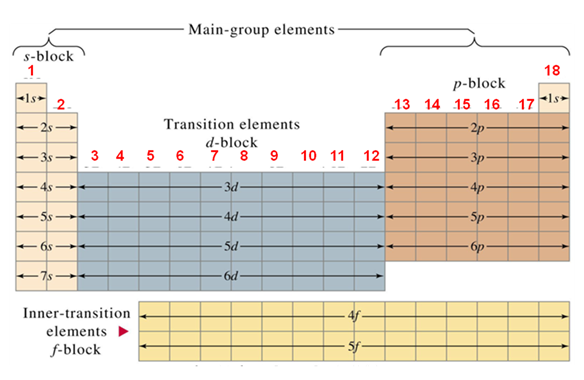
The atomic number is related to the electronic configuration of the elements. The chemical properties of an element are governed by the outer shell electronic configuration. Hence, atomic number and chemical properties are correlated to each other.
The elements having same electronic configuration in the outer most shell should have same chemical properties and hence, they can be placed in a same group.
Elements with one s electron is placed in Group 1 (the alkali metals), with two s electrons is placed in Group 2 (the alkali earth metals). Elements with three electrons (two s and one p ) in the outer shell are called Group 13. On successive addition of electron in p group number increases successively.
In the same way the elements where d orbitals are filled up by successive addition of electron are called d - block elements or transition metal elements. Transition metal elements are situated from Group 3 to Group 12.
Finally elements where f - orbitals are filled up, known as f - block elements or lanthanide and actinide series elements.
It has been stated earlier that chemical property of elements depends on outer shell electronic configuration. The outer shell electrons are ruled by effective nuclear charge. Hence, effective nuclear charge governs the following properties,
(i) Size of atom, atomic volume, atomic radii, and ionic radii
(ii) Ionization potential
(iii) Electron affinity
(iv) Electronegativity
(v) Metallic property