Periodic Table:
In 1869, Russian scientist Drmiti Meldeleev has tabulated then existing elements according to their atomic weight. The table is known as periodic table. According to Mendeleev, physical and chemical properties of elements are periodic functions of their atomic weights. Distributions of elements along a horizontal row forms a period, while, along a vertical column forms a group. He arranged elements into eight main groups and twelve columns according to similarities in physical and chemicals properties´ of the existing elements. Each group was subdivided into two sub groups.
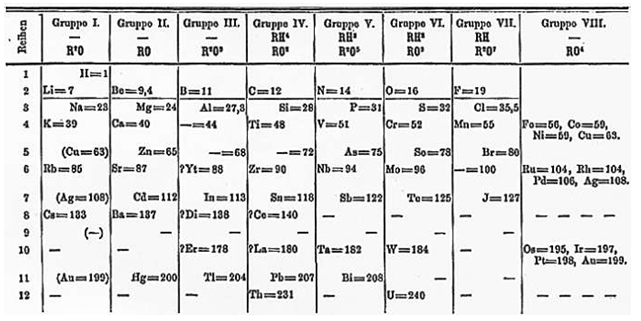
Table. Mendeleev periodic table*:
*http://en.wikipedia.org/wiki/Dmitri_Mendeleev
Merits and limitations of the Mendeleev periodic table:
Merits:
In Mendeleev periodic table, elements were arranged according to their similarities in physical and chemical properties. Therefore, it makes easier for studying and remembering the properties of huge numbers of elements just knowing the properties of the group . Furthermore, based on the periodic table the properties of some unknown elements can be predicted. This was a great achievement.
Limitations:
Hydrogen was placed in Group I, although, its properties resembled the Group I elements (the alkali metals) as well as the group VII elements (the halogens). Hence, in the periodic table the position of hydrogen was not justified.
In some cases Mendeleev placed elements according to their similarities in properties and not in increasing order of their atomic masses, while some dissimilar elements were grouped together. Thus, the position of these elements was not justified. For example, cobalt (at. mass 58.9) was placed before nickel (at. mass 58.6); copper and mercury are similar in their properties but were placed separately. Copper was placed in group I although it did not resemble the elements of this group.
Novel metals, e . g . Cu, Ag, Au were placed in Group I with chemically dissimilar alkaline earth metals. Moreover, Mn has been placed with halogens.
Same element with different atomic mass but same in atomic number is known as isotope of the element. In Mendeleev periodic table isotopes should have different place, but there was no province for the positions of isotopes.
In certain pairs of elements like, Ar (40) and K (39); Co (58.9) and Ni (58.6); Te (127.6) and I (126.9) the arrangement was not justified, e . g . Ar was placed before K.
Valences states or the elements have not been considered for their placement in a group.
Fourteen elements that follow lanthanum called lanthanides and fourteen elements following actinium called actinides were not given proper places in Mendeleev's periodic table, rather they have been provide two rows at the bottom of the table.