4.10.3. Elements of ADCs
ADCs are made up of three components:
- a monoclonal antibody: An important factor in the successful development of ADCs is the selection of wellcharacterized antigens to serve as the target for the antibody. The full expression pattern of the antigen throughout the body and on both healthy and tumor cells must be taken into consideration in order to avoid unwanted toxic side effects. Likewise, the expression of tumor-specific antigens within the tumor itself may be heterogeneous, with some cells displaying the antigen and others not, and this can affect the efficacy of the ADC (Figure 13).
Generally, B- and T-cell surface proteins are frequently chosen as target antigens since they are widely expressed on the surface of malignant B and T cells in these types of cancer.
Targets include B-cell surface proteins such as CD20, CD22, CD40, and CD79, and T-cell surface proteins such as CD25 and CD30, as well as proteins that are overexpressed on carcinoma cells, including the human epidermal growth factor receptor 2 (HER2), epidermal growth factor receptor (EGFR), prostate-specific membrane antigen (PSMA), and Cryptic family protein 1B (Cripto).
- a cytotoxic drug: Three main types of drugs are used as the cytotoxic component of ADCs: calicheamicin, maytansinoids, and auristatins. They fall into two classes, according to the mechanism via which they lead to cell death, with the two latter types causing the unraveling of structural fibers in the cell, and calicheamycin causing irreparable DNA damage. In order to maximize killing potential, the cytotoxic agent needs to be highly potent. Using highly toxic drugs that are more potent than standard chemotherapy agents ensures that what little gets into the cell has the maximum effect (Figure 13).
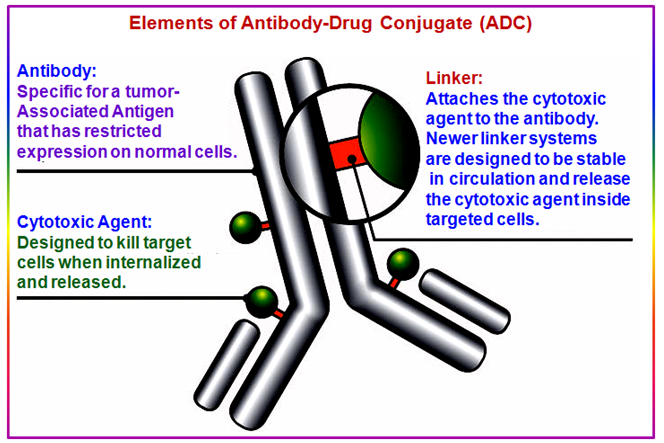
- a linker: The linker joins the antibody and drug together. The linker region is extremely important to ADC design, as a delicate balance must be struck between stability in the bloodstream (to ensure the toxin is not released prematurely) and efficient release inside the cell once it entered.
Cleavable or releasable linkers that are broken down inside the cell are chosen to allow drug release inside the cell. (i) Initially, hydrazone linkers (-C=N-NH-) which are broken down only in the acidic environment of the cell were used. However, these often suffer from poor stability in the blood. Thus, the antibody falls off and the ADC fails to strike multiple targets around the body. Hence poor stability of linker renders the ADC ineffective or causes toxic side effects (Figure 13).
Figure 13. Elements of antibody-drug conjugates.
(ii) A range of linker that improves the stability of the ADC, while still allowing efficient drug release in the tumor cell has been development. These include disulfide-based linkers (-S-S-) that are selectively broken down inside the tumor cell where the concentration of thiols is higher than in the blood and peptide linkers (-NH-CO-) that are broken down by enzymes found inside the tumor cell.
(iii) Non-cleavable linkers like thioether (-S-) linkers are being used now a days after the discovery that the cleavage of linker is not necessary in order for the drug to become activated. It is now discovered that the linker can be degraded by the cellular protein degradation machinery, leading to drug activation.
(iv) Another linker type that was recently described is the maleimidyl-based hydrophilic linker (PEG4Mal linker), which is designed to target drug-resistant tumor cells as it cannot be pumped through multidrug resistance channels on the cell surface.
To make an effective ADC the following key factors must be kept in mind:
- A target antigen, which is cancer specific
- A monocolonal antibody, diabody or ScV fragment that displays a high binding affinity to the antigen
- A cytotoxic agent that is highly potent
- linker designed to allow ADCs to remain inactive when in the blood