4.10. Antibody–Drug Conjugates
4.10.1. Origins of M onoclonal A ntibody T herapy
Traditional chemotherapy is the pillar for cancer treatment. However, cytotoxic agents are not tumour specific. The rapidly proliferating cells are more prone to the cytotoxic effect of these drugs. Therefore, increased toxicities of such chemotherapeutic cytotoxic agents against normal tissues are major drawback of Traditional chemotherapy of cancer. As a consequence, anticancer chemotherapeutics are often given at suboptimal doses.
Therefore, searching for an alternative tumor specific agents to kill tumor cell gave rise to the concept of immunotherapy which takes advantages of the immune systems. The immune system responds to the environmental factors it encounters on the basis of discrimination between self and non-self. Tumor cells are not specifically targeted by one's immune system since tumor cells are the patient's own cells. Tumor cells, however are highly abnormal, and many display unusual antigens that are either inappropriate for the cell type, its environment, or are only normally present such as fetal antigens during the organisms' development.
Other tumor cells display cell surface receptors that are rare or absent on the surfaces of healthy cells, and which are responsible for activating cellular signal transduction pathways that cause the unregulated growth and division of the tumor cell. Examples include ErbB2, a constitutively active cell surface receptor that is produced at abnormally high levels on the surface of approximately 30% of breast cancer tumor cells. Such breast cancer is known a HER2 positive breast cancer.
Immunotherapy developed as a technique with the discovery of the structure of antibodies and the development of hybridoma technology, which provided the first reliable source of monoclonal antibodies (mAb) which are a key component of the adaptive immune response and playing a central role in both in the recognition of foreign antigens and the stimulation of an immune response to them. The advent of monoclonal antibody technology has made it possible to raise antibodies against specific antigens presented on the surfaces of tumors, thereby allowing specific targeting of tumors both in vitro and in vivo. Initial research on malignant neoplasms found mAb therapy of limited and generally short-lived success with malignancies of the blood. Furthermore treatment had to be specifically tailored to each individual patient, thus proving to be impracticable for the routine clinical setting.
Throughout the progression of therapeutic monoclonal antibody drug development there have been four major antibody types developed: murine, chimeric, humanised and human. Initial therapeutic antibodies were simple murine analogues, which contributed to the early lack of success. It has since been shown that these antibodies have: a short half-life in vivo (due to immune complex formation), limited penetration into tumour sites, and that they inadequately recruit host effector functions. To overcome these difficulties the technical issues initially experienced had to be surpassed. Chimeric and humanized antibodies have generally replaced murine antibodies in modern therapeutic antibody applications. Hybridoma technology has been replaced by recombinant DNA technology, transgenic mice and phage display.
In the past few years, more than 30 immunoglobulins (IgGs) and their derivatives have been approved for use in various disease indications. Many antibody-based drugs have been approved and marketed to treat diseases such as cancer and inflammatory diseases that are affecting large numbers of patients. They are also being used for more specialized indications owing to special regulatory procedures for rare medical conditions like paroxysmal nocturnal haemoglobinuria Currently, 26 antibodies are in Phase III clinical trials (35%) and 9 out of them have got orphan drug designation.
A detailed knowledge of antibody structure and activity allows researchers to engineer them on a more rational way for improving their pharmaceutical properties, target specificity, stability, and functional potency. Therefore, many c linically useful unconjugated monoclonal antibodies (mAbs) have been developed which can selectively recognize antigens that are preferentially expressed on or near tumor cells. These antibodies exert their cytotoxic effects through mechanisms such as cell signaling, antibody-dependent cellular cytotoxicity, antibody-dependent cellular phagocytosis, and complement-dependent cytotoxicity. However, the majority of these mAbs are used in combination with chemotherapy, and many others have demonstrated insufficient clinical activity. Therefore, significant efforts have been put forth to gear up mAbs through various modifications towards enhancing their functional diversity, ability to kill tumor cell with high potency and target specificity. One approach by which the activities of these mAbs have been enhanced is through conjugation with cytotoxic drugs, generating antibody-drug conjugates (ADCs) capable of antigen-specific delivery of highly potent cytotoxic drugs to tumor cells.
In summary, anti-neoplastic drugs like doxorubicin, daunomycin, vinca-alkaloids, and taxoids etc. have demonstrated their ability to kill cancer cells but generally with limited selectivity and high toxic effects on normal cells yielding marginal therapeutic indices. On the other hand, approved naked antibody drugs like rituximab, trastuzumab, cetuximab, bevacizumab, panitumumab, alemtuzumab, and ofatumumab have demonstrated their therapeutic utility in malignancies. However it is observed that these mAbs often show significant clinical efficacy when linked with small cytotoxic drugs. Therefore, covalent conjugation of mAbs and drugs with synthetic chemical linkers to create antibody-drug conjugates is though an older concept (In the 1960s ADCs was used in animal models and in the 1980s, clinical trials with murine IgG-based ADCs were conducted) but now a day widely used for treating tumor.
4.10.2. Defining Antibody-Drug Conjugates: ADC Technology
Antibody Drug Conjugates (ADCs) are therapeutic agents designed to target the delivery of a cytotoxic drug specifically to tumor cells ( Figure 12 ). In ADCs cytotoxic agents are linked to monoclonal antibodies that bind to tumor cell-specific antigens or to antigens that are overexpressed on the surface of tumor cells. The antibody acts as a sort of GPS system and it increases delivery of potent cell-killing drugs to the tumor cell and reduces the exposure of normal cells.
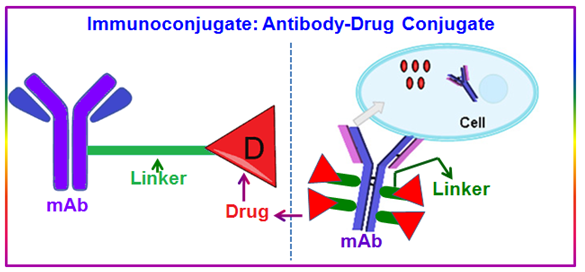
Figure 12. Shematic drawing of antibody-drug conjugates.