3.6.5 Cobalt Catalysts
Cobalt is present in various metalloenzymes like methylmalonyl coenzyme A mutase. It is also a key constituent of vitamin B12 which plays an important role in normal functioning of the central nervous system. Cobalt exists mainly in two oxidation states. The standard electrode potential for Co(III)/Co(II) system is 1.82 V which shows that it is a very strong oxidant. Also, the existence of cobalt based redox systems in biological systems has intuited the exploration of similar bio-mimicking systems for redox reactions. Some of these reactions are illustrated below.
3.6.5.1 Oxidation Reactions
Epoxidation of alkenes can be achieved by using cobalt(II)-Schiff base complex in the presence of reducing agent such as aldehydes, β-ketoester, acetal and triphenylphosphine under molecular oxygen (Scheme 1). A series of cobalt(II) complexes have been designed and developed for this purpose. The reactions are general, efficient and effective at ambient temperature.
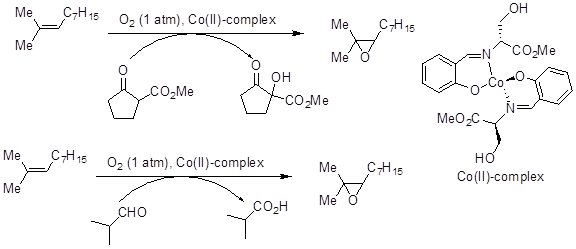
Scheme 1