3.6.4.5.2 Sulfoxidation
The oxidation of aryl alkyl sulfides has been accomplished using in situ generated Fe(III)-Schiff base complex in the presence of 30% H2 O2 as terminal oxidant (Scheme 8). Addition of benzoic acid as an additive enhances the enantioselectivity significantly.
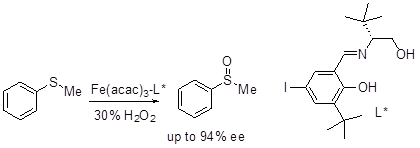
Scheme 8
3.6.4.5.3 Cycloaddition
Chiral iron(III)- bis (oxazoline) has been used for the asymmetric (4+2)-cycloaddition reaction with good enantioselectivity (Scheme 9).
Scheme 9