3.6.4 Iron Catalysts
Iron is 3d -series transition element belonging to Group 8. It is present in haemoglobin and myoglobin and so it is one of the most important metals in biology. It exists in +2 and +3 oxidation states mainly. In +3 oxidation state it is a strong Lewis acid while metallic iron may behave as a single electron donor under suitable conditions.
3.6.4.1 Reduction of Nitro Group
Aromatic nitro compounds can be reduced to the corresponding aromatic amines in presence of iron powder under acidic conditions. The reaction proceeds through single electron transfer in similar lines to Clemensen reduction (Scheme 1).
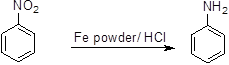
Scheme 1
3.6.4.2 C-C Bond Formation
The Lewis acidity of iron(III) has been utilized for several reactions. These reactions mostly involve activation by co-ordination with heteroatoms or π-bonds. Bromination of aromatic compounds can be carried out by bromine in the presence of iron powder. Similarly, addition of catalytic amount of iron(III) salts facilitates the addition of organometallic nucleophiles (Scheme 2).
Cationic [(allyl)Fe(CO)4] is known to react with different types of nucleophiles regioselectively and stereoselectively. If the reaction is carried out at low temperature then the olefin geometry can be preserved during substitution as the π-σ-π-isomerization is slow in this case. If allyl substrates possess electron withdrawing group, then nucleophillic substitution of the leaving group proceeds regioselectively (Scheme 3).
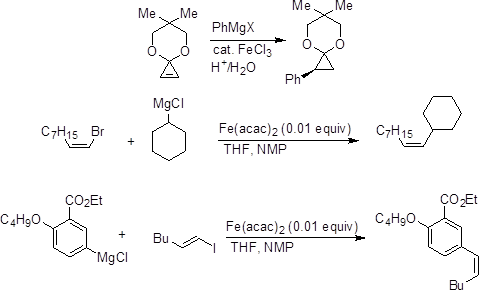
Scheme 2