4-2: Organic wastes
(1) Waste waters and sewage sludge:
(2) Animal manures:
Our nation produces more than one billion Mg of solid manure annually, or 4.5 Mg per capita.
"Modern" manure often contains higher salts (~10%), antibiotics, and metals.
Manure has been one of the major sources of nutrient pollution for many water systems, especially near feedlots.
Agricultural and Human runoff
(example of non-point source pollution)
- Excessive nutrients
- loss from the soil
- eutrophication downstream
- health hazards- nitrate in groundwater = 1000x risk of methemoglobinemia (a blood disorder)
- Toxic and chemicals
- particularly heavy metals and salts
- pesticides, herbicides, etc
- pathogens (cholera, typhoid, etc)
- hormones, antibiotics
“Xenobiotic” – a compound that shows up where it
biologically shouldn't
- human hormones in fish downstream; dioxins
4-3. Heavy metals (Hg, Pb, Cd, Cr,Ni, etc.)
Sources: industrial wastes, mines, agricultural inputs (P fertilizers, sludge, etc), the use of coal and other mined materials. They can be toxic to plants and animals. Many of them bioaccumulate as they move around in ecosystems. They are very immobile in soils, therefore, difficult to remove.
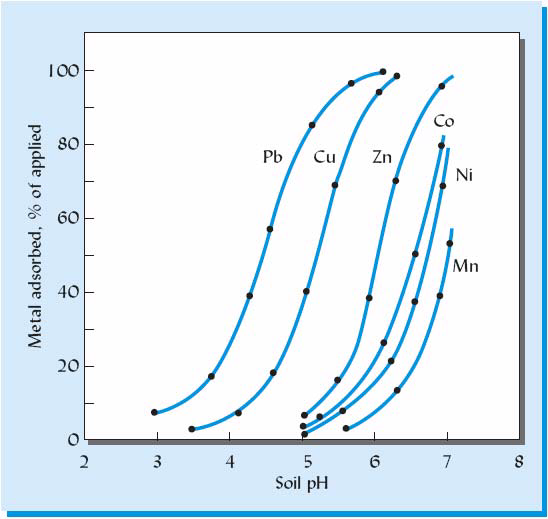
The effect of pH on the adsorption of six heavy metals. The metals were adsorbed by clay-sized goethite (an iron oxide mineral) that forms coatings on many soil particles. Maintaining the soil pH near 7 (neutral) is expected to maximize the adsorption and thereby minimize the solution concentration of most heavy metals, especially of copper and lead. [Modified from Basta et al. (2005)]