Preliminary treatment processes
- Grit removal
- Sedimentation and Clarification
- Removal of colloidal suspensions by Coagulation
- Flow through Porous Media
Chemical treatment
A number of constituents of waters and wastewaters do not respond to the conventional treatment processes already discussed
Chemical precipitation
Removal of certain soluble inorganic materials can be achieved by the addition of suitable reagents to convert the soluble impurities into insoluble precipitates which can then be flocculated and removed by sedimentation.
The extent of removal which can be accomplished depends on the solubility of the product; this is usually affected by such factors as pH and temperature.
By the addition of ferrous sulphate and lime the chromium is reduced to the trivalent form which can be precipitated as a hydroxide
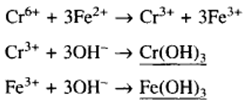
For chromium reduction the theoretical requirement is shown above, but at this level the reaction proceeds very slowly and in practice, to ensure complete reduction, it is necessary to add 5-6 atoms of ferrous iron for each atom of hexavalent chromium.
Phosphates can be precipitated from solution by the addition of metal ions, a process used for the removal of nutrients from wastewater effluents
A characteristic of chemical precipitation processes is the production of relatively large volumes of sludge.
A common use of chemical precipitation is in water softening.