Another group of ozone direct reactions are those with inorganic species such as Fe2+, Mn2+, NO2-, OH-, HO2-, etc.
These could be defined as redox reactions because in the overall process ozone acts as a true oxidizing agent by taking electrons whereas the other species act as true reducing agents by losing electrons.
Ozone has the highest standard redox potential among conventional oxidants such as chlorine, chlorine dioxide, permanganate ion, and hydrogen peroxide
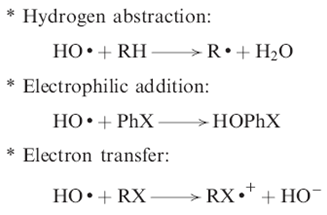
The indirect type of ozonation is due to the reactions of free radical species, especially the hydroxyl radical, with the organic matter present in water.
These free radicals come from reaction mechanisms of ozone decomposition in water that can be initiated by the hydroxyl ion or, to be more precise, by the hydroperoxide ion as shown in reactions
Ozone reacts very selectively through direct reactions with compounds with specific functional groups in their molecules.
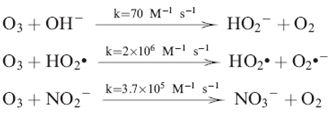
Mechanism of O3 decomposition in Water
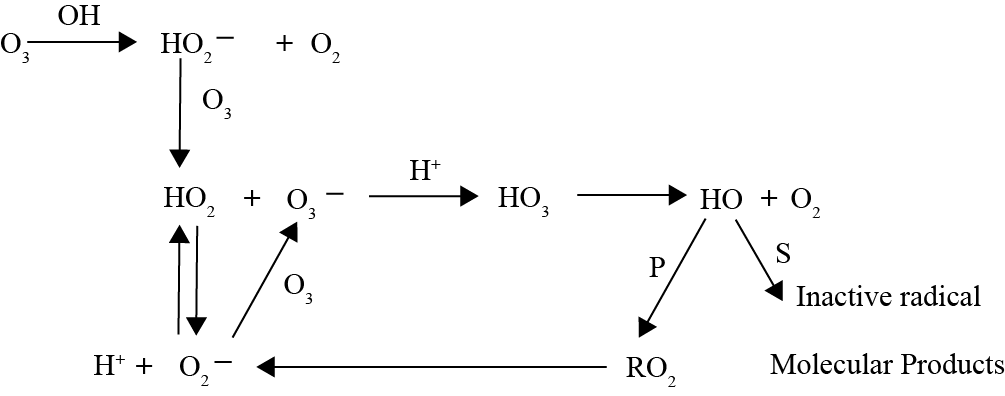
Hydrogen Peroxide Oxidation
Similar to ozone, hydrogen peroxide can react with organic matter present in water through direct and indirect pathways. In direct mechanisms, hydrogen peroxide participates in redox reactions where it can behave as an oxidant or as a reductant:
Combined Oxidations: O3/H2O2, UV/H2O2, and O3/UV
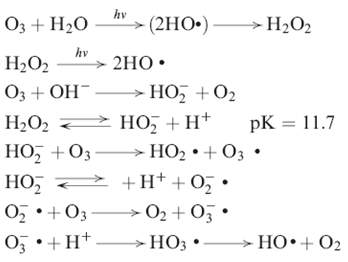
Among free radicals, the hydroxyl radical shows a high oxidizing power, and it is generally accepted as the main oxidant in these advanced oxidation systems.