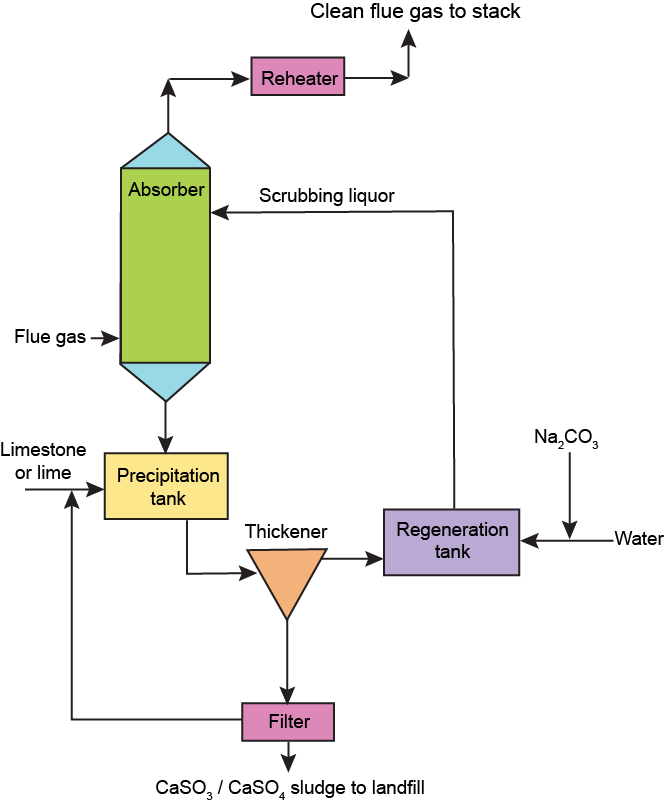
In recent years, a new scrubber design that uses lime (calcium hydroxide; Ca(OH)2) rather than limestone has been introduced in many plants
![]()
Lime is more chemically reactive than limestone, so the amount needed to remove sulfur dioxide from fl ue gases is comparably less.
Other basic compounds have been used in scrubbers for the removal of sulfur dioxide.
Among the most promising are amines.
Amines react readily with sulfur dioxide to form a salt.
When the salt is treated with steam (a process known as steam stripping), the sulfur dioxide dissolves and is removed and the amine is regenerated.
The amine can then be reused in another cycle of sulfur dioxide removal.
Limestone Scrubbing